Usman Baber*, MD, MS
AsiaIntervention 2019;5:9-11, DOI: 10.4244/AIJV5I1A2
* Cardiovascular Institute, Icahn School of Medicine at Mount Sinai, New York, USA
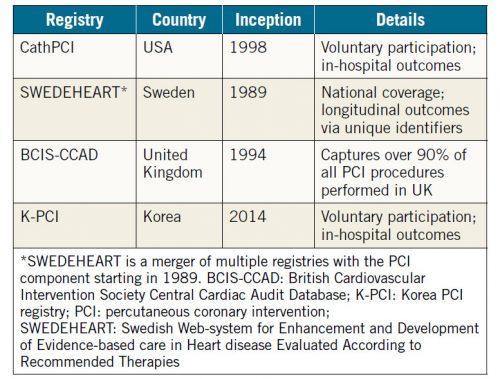
Leveraging “real-world” data via administrative claims, registries or other curated sources is increasingly utilised to inform clinical practice, characterise disease epidemiology and guide public policy. The digitisation of medical records, coupled with rapid advances in computational speed and sophistication, has enabled this electronic transition. Resources such as the United States Center for Disease Control (CDC) National Health and Nutrition Examination Survey (NHANES) provide ongoing and representative estimates of various health, nutrition and fitness metrics for the US adult population. In contrast, dedicated registries focused on disease or procedural domains provide temporal trends on healthcare resource utilisation, variation in practice patterns and consequences of policy or guideline implementation (Table 1). In certain instances, registries are increasingly utilised to facilitate the conduct of pragmatic clinical trials that increase study efficiency, and reduce costs and time to trial completion1. These data also enable the evaluation of aetiologic associations between various exposures and disease epidemiology, which may be discerned more reliably from real-world as opposed to clinical trial cohorts2. One illustration of this concept was the seminal paper by Go et al demonstrating the prevalence and impact of renal impairment on cardiovascular outcomes using administrative claims data from a million healthcare plan participants3. Subsequent reports have confirmed these earlier observations in both primary and secondary prevention settings, and chronic kidney disease (CKD) is now included as a risk-enhancing feature that warrants a more aggressive approach towards risk factor modification and control of blood lipids4. Nevertheless, the evidence base for CKD and related prognosis is primarily derived from Western European and North American populations comprised of European or African ancestry. Indeed, validated equations that provide estimates of glomerular filtration rate (eGFR) include terms for self-described African or non-African race5,6. Analogous data evaluating similar associations among individuals in lower middle-income countries in the Middle East or other non-Western regions remain limited.
Table 1. Selected national PCI registries.
To address this gap, Hammoudeh et al examined the prevalence, clinical profile and impact of CKD using data from the first, national all-comer Jordanian PCI registry (n=2,426)7.
Despite a relatively young mean age of only 56 years, the prevalence of reduced eGFR (<90 ml/min) was 60%, with approximately 14% of patients presenting with at least moderate to severely reduced kidney function (eGFR <60 ml/min). Not surprisingly, patients with more advanced CKD were characterised by older age and more comorbidities including hypertension, diabetes mellitus and prior revascularisation. Discharge pharmacotherapy, including use of beta-blockers and statins, did not vary substantially in relation to renal function. With respect to outcomes, in-hospital bleeding was relatively uncommon yet higher among those with the most severe renal impairment, a finding entirely consistent with the well-known association between CKD and bleeding risk8. Similar numerical patterns were observed at 30 days and one year, albeit not statistically significant due to limited power. With regard to ischaemic events, CKD emerged as an independent correlate of cardiovascular death, while differences in rehospitalisation for acute coronary syndrome (ACS) and revascularisation did not vary by renal function.
An intriguing result from the present report is the excess risk for cardiovascular death observed among those with CKD in the absence of a concordant rise in ACS hospitalisation, which may serve as a crude surrogate for non-fatal myocardial infarction (MI). Indeed, this finding is consistent with the transition in cardiovascular epidemiology that occurs as renal function worsens. Specifically, while atherosclerosis-mediated mechanisms modulate cardiac risk in mild CKD, vascular calcification, myocardial fibrosis and electrical instability emerge as contributors at more advanced levels of renal impairment9,10. The numerical trend suggesting more frequent heart failure hospitalisation with worsening CKD in the current study is also consistent with this paradigm. Hence, treatments that target the unique vascular phenotype present in moderate to advanced CKD11 may provide greater benefit than established therapies commonly used to treat atherosclerosis in the absence of renal dysfunction.
The authors contend that PCI is safe and effective among those with CKD and that an invasive strategy should be considered more frequently in such patients. While this may be true, inferential claims based upon observational data should be interpreted cautiously. In addition, the relatively low rate of adverse events may have introduced a type II error when comparing across different levels of renal function. In contrast, stent thrombosis occurred relatively frequently (~1.5-2%), a substantially higher rate in comparison to other contemporary registries12. This is a concerning result, and more details on stent type and one-year adherence would be insightful, along with the type of P2Y12 inhibitor prescribed at discharge. More granularity for these data elements would certainly be welcome in future reports from this registry.
Notwithstanding these limitations, the authors and investigators leading JoPCR1 should be commended for these efforts. In addition to evaluating outcomes in a broad population of Jordanian patients undergoing PCI, this resource will provide opportunities for data sharing, academic collaboration and identification of risk factors and treatment approaches unique to the Middle East versus other regions. As with any registry-based product it will be important to implement processes, such as periodic auditing, to ensure that data are valid, reproducible and high-quality. Representing the latest entry into the global market of outcome-oriented PCI registries, additional insights from JoPCR1 are eagerly awaited.
Conflict of interest statement
The author has received speaker’s fees from AstraZeneca and Boston Scientific.
References
1. Rao SV, Hess CN, Barham B, Aberle LH, Anstrom KJ, Patel TB, Jorgensen JP, Mazzaferri EL Jr, Jolly SS, Jacobs A, Newby LK, Gibson CM, Kong DF, Mehran R, Waksman R, Gilchrist IC, McCourt BJ, Messenger JC, Peterson ED, Harrington RA, Krucoff MW. A registry-based randomized trial comparing radial and femoral approaches in women undergoing percutaneous coronary intervention: the SAFE-PCI for Women (Study of Access Site for Enhancement of PCI for Women) trial. JACC Cardiovasc Interv. 2014;7:857-67.
2. Ho PM, Peterson PN, Masoudi FA. Evaluating the evidence: is there a rigid hierarchy? Circulation. 2008;118:1675-84.
3. Go AS, Chertow GM, Fan D, McCulloch CE, Hsu CY. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N Engl J Med. 2004;351:1296-305.
4. Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, Braun LT, de Ferranti S, Faiella-Tommasino J, Forman DE, Goldberg R, Heidenreich PA, Hlatky MA, Jones DW, Lloyd-Jones D, Lopez-Pajares N, Ndumele CE, Orringer CE, Peralta CA, Saseen JJ, Smith SC Jr, Sperling L, Virani SS, Yeboah J. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APHA/ ASPC/NLA/PCNA Guideline on the Management of Blood Cholesterol: Executive Summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2018 Nov 3. [Epub ahead of print].
5. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D. A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med. 1999;130:461-70.
6. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-12.
7. Hammoudeh A, Al-Bayyari N, Obeidat O, Al-Mousa E, Tabbalat R, Alhaddad I. Incidence and impact on prognosis of impaired kidney function in Middle Eastern patients undergoing percutaneous coronary intervention: results from the first Jordanian PCI registry. AsiaIntervention. 2019;5:18-26.
8. Baber U, Mehran R, Giustino G, Cohen DJ, Henry TD, Sartori S, Ariti C, Litherland C, Dangas G, Gibson CM, Krucoff MW, Moliterno DJ, Kirtane AJ, Stone GW, Colombo A, Chieffo A, Kini AS, Witzenbichler B, Weisz G, Steg PG, Pocock S. Coronary Thrombosis and Major Bleeding After PCI With Drug-Eluting Stents: Risk Scores From PARIS. J Am Coll Cardiol. 2016;67: 2224-34.
9. Hakeem A, Bhatti S, Dillie KS, Cook JR, Samad Z, Roth- Cline MD, Chang SM. Predictive value of myocardial perfusion single-photon emission computed tomography and the impact of renal function on cardiac death. Circulation. 2008;118: 2540-9.
10. Herzog CA, Shroff GR. Atherosclerotic versus nonatherosclerotic evaluation: the Yin and Yang of cardiovascular imaging in advanced chronic kidney disease. JACC Cardiovasc Imaging. 2014;7:729-32.
11. Matsumoto Y, Mori Y, Kageyama S, Arihara K, Sugiyama T, Ohmura H, Yakushigawa T, Sugiyama H, Shimada Y, Nojima Y, Shio N. Spironolactone reduces cardiovascular and cerebrovascular morbidity and mortality in hemodialysis patients. J Am Coll Cardiol. 2014;63:528-36.
12. Stone GW, Witzenbichler B, Weisz G, Rinaldi MJ, Neumann FJ, Metzger DC, Henry TD, Cox DA, Duffy PL, Mazzaferri E, Gurbel PA, Xu K, Parise H, Kirtane AJ, Brodie BR, Mehran R, Stuckey TD; ADAPT-DES Investigators. Platelet reactivity and clinical outcomes after coronary artery implantation of drug-eluting stents (ADAPT-DES): a prospective multicentre registry study. Lancet. 2013;382:614-23.
To download, please click below.