Introduction
Coronary artery disease (CAD) due to atherosclerosis is projected to be the leading cause of morbidity and mortality worldwide until 20401. CAD affects approximately 2.6 million people in the United Kingdom (UK) and 1 in 4 of them do not experience any symptoms2. Sudden rupture or erosion of a previously undiagnosed atherosclerotic plaque leads to acute thrombosis of a coronary artery (termed acute coronary syndrome [ACS]), and potentially, sudden death. Early diagnosis of atherosclerosis is important in order to prevent myocardial infarction (MI) in patients at risk.
Initial arterial changes in early atherosclerotic disease include compensatory enlargement of both the outer wall of the vessel as well as the lumen, known as positive remodelling (PR). PR is aimed at delaying the development of significant lumen compromise; hence, it is beneficial in preventing loss or reduction of coronary flow. However, PR is an important feature that has been associated with extension of the lipid necrotic core and, thus, plaque vulnerability3. Coronary artery plaques that undergo PR have larger lipid cores, which may be an explanation for the paradox of PR – it is both useful in avoiding luminal stenosis but also harmful as it promotes plaque vulnerability4. These findings explain the common occurrence of plaque rupture at sites of significant plaque accumulation related to PR5.
Computed tomography coronary angiography (CTCA) has emerged as a precise and robust non-invasive imaging tool, which facilitates the assessment of CAD. CTCA has almost 100% accuracy in excluding significant CAD and, when compared with functional imaging tests and invasive angiography, is the cheapest technique6. This highly sensitive imaging modality is increasingly used to assess patients presenting with stable chest pain, and, in 2016, the National Institute for Health and Care Excellence (NICE), which provides evidence-based guidelines for England’s National Health Service (NHS), recommended CTCA as the first-line investigation tool for patients with chest pain due to suspected CAD7.
CTCA is useful in identifying coronary arteries with features related to high-risk plaque (HRP), namely spotty calcification, low-attenuation plaque, napkin-ring sign, and PR3. In the absence of severe calcification, CTCA enables accurate measurement of vessel size and, thus, of PR pattern8.
We aimed to assess the characteristics and outcomes of patients from our Rapid Access Chest Pain Clinic (RACPC) who were referred for CTCA with plaque features of PR on their CTCA images. The primary endpoint was the composite of ACS and revascularisation.
Methods
Patient selection
A total of 2,205 patients who were referred for CTCA from June 2018 to January 2020 in Sandwell and West Birmingham Hospitals NHS Trust were retrospectively identified (Figure 1). We excluded non-RACPC referrals, duplicate records, patients whose CTCA images were unable to be extracted from the archive, patients with previous coronary artery bypass graft (CABG), normal coronaries (as defined by the Coronary Artery Disease – Reporting and Data System [CAD-RADS] 0=normal coronary stenosis) or minimal CAD (as defined by the CAD-RADS 1=1-24% coronary stenosis), and degraded images9.
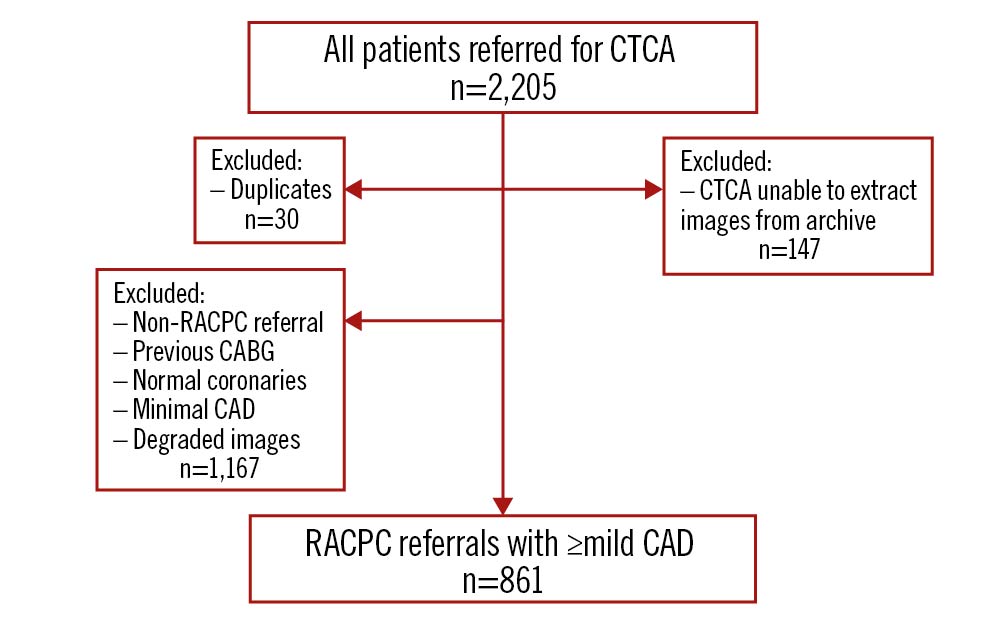
Figure 1. Patient selection. CABG: coronary artery bypass; CAD: coronary artery disease; CTCA: computed tomography coronary angiogram; RACPC: Rapid Access Chest Pain Clinic
CTCA protocol
Baseline observations were performed, including heart rate and blood pressure, as part of the CTCA checklist. A venous cannula (minimum 18G) was preferably inserted in the ante-cubital fossa of the patients and standard electrocardiogram (ECG) leads were placed. After confirmatory checks, the patients were given sublingual glyceryl trinitrate (GTN) spray (400 μg) or a GTN tablet (500 μg) and intravenous metoprolol (maximum 50 mg). Breath-holding was practised.
Patients underwent prospective, gated CTCA with a 128-slice dual-source computed tomography (DSCT) scanner (Somatom Definition Flash; Siemens). Based on the patient’s body mass index (BMI), the tube voltage (<25 kg/m2: 100 kV, ≥25 kg/m2: 120 kV) and current were adjusted. When a standard adapt (sequential scan) was performed, single-source 128×0.6 mm acquisition was utilised with a rotation time of 0.28 seconds. This was performed at 70% of the standard cardiac cycle. The systolic scan (also an adapt scan) had additional padding and was performed at 250-350 ms of the cardiac cycle.
The contrast bolus (at a rate of 4.5ml/s with a high-pressure injector) for the main scan was as follows:
- 10 ml saline
- 60 ml Iomeron 400 (contrast)
- 40 ml saline
An automated multilingual voice recording was incorporated to instruct breath-holding during the scan. Imaging was performed from 2 cm below the tracheal bifurcation to the level of the diaphragm in a cranio-caudal manner. The data were rebuilt, including post-processing for maximum intensity projection (MIP), multiplanar reformation (MPR), as well as volume rendering, and transferred automatically to the workstation (syngo.via; Siemens).
CT coronary angiographic measurements and definition
PR is defined by a remodelling index (RI) of ≥1.1, which is calculated by the vessel size at the site of the maximal remodelling divided by the vessel size of the reference site10. An RI of ≥1.1 has been identified as the optimal threshold to identify PR, when compared with intravascular ultrasound (IVUS)10.
PR was visually assessed in multiplanar reconstructed and reformatted images, both in the long and short axes of the coronary arteries. Additional manual measurements were performed at the readers’ discretion and a threshold of 1.1 was used to define PR. Operators assessing the CTCA images were level 2 CTCA-trained and had at least 1 year of experience in reporting CTCA images.
Statistical analysis
Statistical analysis was performed using GraphPad Prism version 9.1 (GraphPad Software). Continuous variables were presented as mean±standard deviation and categorical variables were presented as percentages. Continuous variables were compared using Student’s unpaired t-test, and in cases of non-normal distribution, the Mann-Whitney U test was used. Categorical variables were compared using Fisher’s exact test. A p-value of 0.05 was considered significant. Interobserver variability for the analysis of HRP (per coronary artery and per type of HRP) was assessed by Cohen’s kappa (κ value)1112. Binary nominal regression analysis was performed with significant univariate variables to identify precursors of the primary endpoint (composite of ACS and revascularisation).
Results
A total of 861 patients from the RACPC referrals were included in our study; 241 (28%) had PR on CTCA, and 620 (72%) had no PR. The overall mean age of our cohort was 63 years. 42% of our patients were male and 38% were of Asian ethnicity. More than half the patients had hypertension (58%), hypercholesterolaemia (57%) and family history of CAD (53%). Of our study group, 34% were diabetics and 47% were smokers. The average BMI was 29.9 kg/m2.
Interobserver variability was performed for 10% of the dataset (n=86) and kappa=0.4 (fair) for PR.
Comparing patients who had features of PR on CTCA and those without PR, there were significant differences in age, gender and history of previous angiography (p<0.05). Table 1 shows the patients’ clinical characteristics and laboratory data. 36% (n=87) had a PR segment in more than 1 artery although only 2 of these patients had a subsequent ACS. The left main stem was the site of PR in 1.8% of patients (n=12), either alone or in conjunction with the left anterior descending artery (LAD).
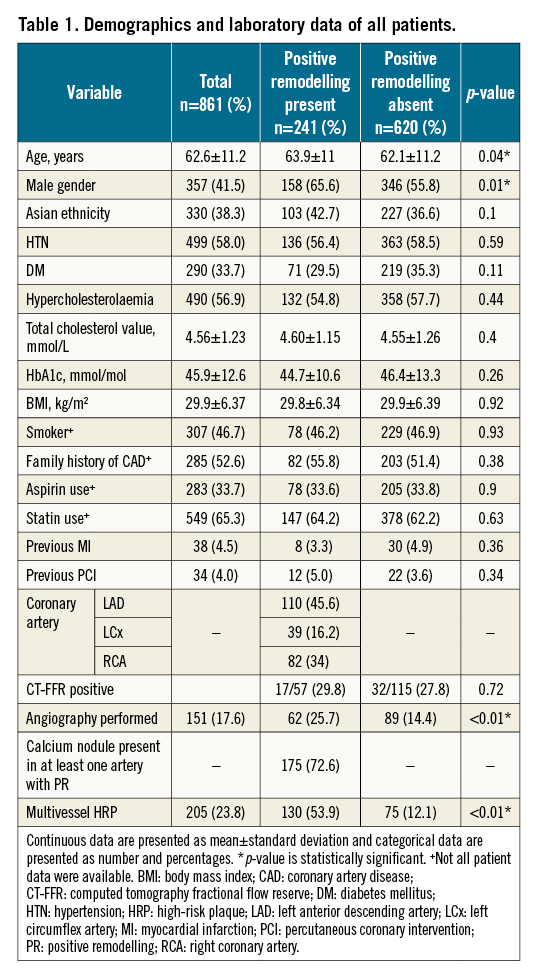
There were also significant increases in subsequent ACS events and the need for revascularisation therapy of either percutaneous coronary intervention (PCI) or CABG (p<0.01) in patients who had PR compared to those without. There was no difference in all-cause mortality. Figure 2 and Table 2 show the outcomes of all patients at 2-year follow-up.
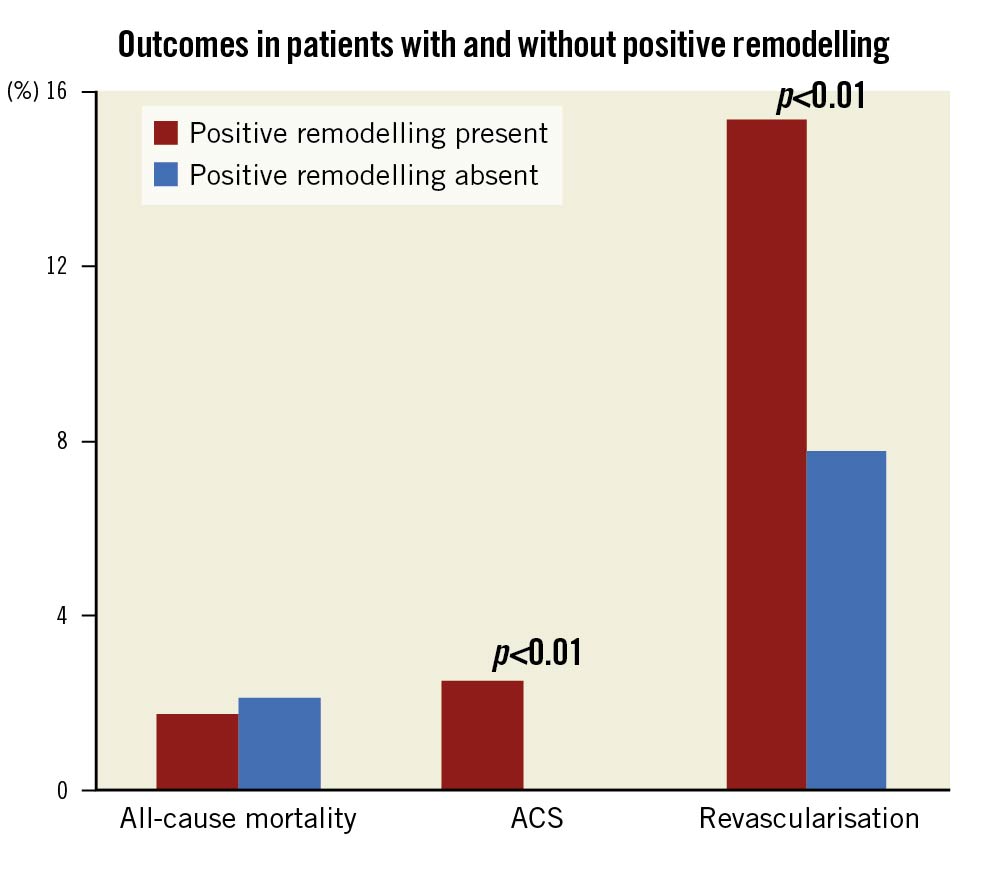
Figure 2. Graphical representation of outcomes in patients with and without positive remodelling. ACS: acute coronary syndrome
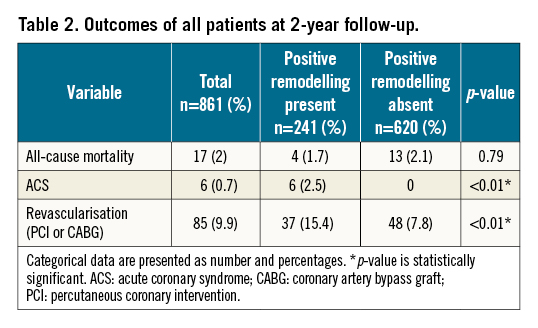
Interestingly, the 6 patients who had readmission with ACS were of male gender and all but one demonstrated PR in their LAD. Binary regression analysis identified age, gender and PR as significant predictors of the composite endpoint of ACS or revascularisation (Figure 3).
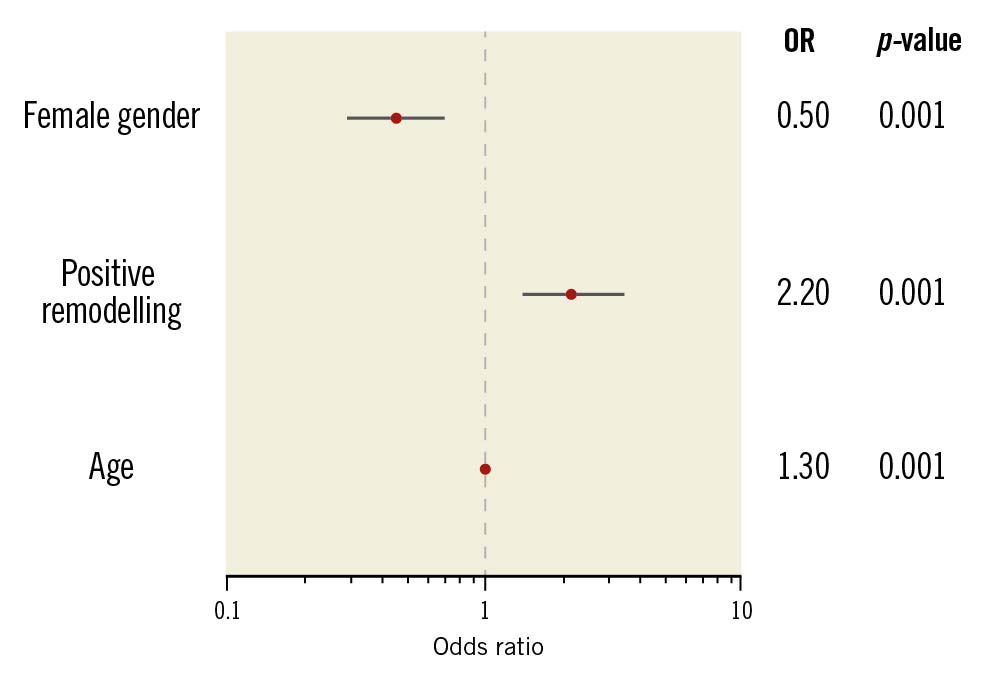
Figure 3. Odds ratio (OR) for the composite endpoint.
Discussion
Our observational study has demonstrated that patients with positive remodelling were of older age and male gender. PR is also of prognostic value in ascertaining which patients are at risk of readmission with ACS and may benefit from revascularisation therapy. This is one of the first studies to identify the characteristics of patients with PR and the subsequent outcomes.
Age and male gender
Increased age and male gender are established risk factors of CAD2. Ageing has been linked with cellular oxidative stress and endothelial dysfunction, both of which are conducive to the progression of PR, as indicated in experimental studies13. These pathophysiological progressions, coupled with other intrinsic factors, such as increased vascular stiffness, thrombogenicity and inflammation explain the susceptibility of older patients to CAD13. Similar to our findings, Burke and his colleagues harvested the hearts of 36 patients who died from severe CAD and found a higher prevalence of PR in males of older age groups1415.
The differences in the incidence and prevalence of ACS between men and women suggest an underlying sex-specific pathophysiology of CAD. Coronary sites with considerable plaque accumulation linked to PR are predisposed to plaque rupture, which has been more commonly reported as a cause of ACS among men5. This is again elucidated by our study, which has shown that females are less likely to have PR. Other infrequent causes of ACS that are more prevalent in women than men include coronary vasospasm, spontaneous coronary artery dissection and stress-related cardiomyopathy, collectively called MI with non-obstructive coronary arteries (MINOCA)16.
Use of statinS and aspirin
Dyslipidaemia, a well-established cardiovascular risk factor, has been associated with intrinsic pathological factors conducive to CAD, including endothelial cell dysfunction, decreased nitric oxide (NO) bioavailability, increased oxidative stress and marked inflammatory response17. Earlier studies have demonstrated that lipid-lowering therapy is associated with vascular remodelling, as an adaptive process to compensate for the decrease in lumen area in patients with hypercholesterolaemia18.
The paradoxical benefit and process of PR could be modulated by statin therapy; the PARADIGM (Progression of Atherosclerotic Plaque Determined by Computed Tomographic Angiographic Imaging) study followed 1,255 patients and found that moderate- to high-intensity statins successfully reduced the risk of developing HRP and positive arterial remodelling19. A subgroup analysis of the 6 patients who developed ACS found that, although they had been prescribed statins post-CTCA, they were not high-intensity statins.
The underlying mechanism of statins in modulating PR remains unclear, but it is likely due to a combination of factors that affect the vessel wall structure and endothelial function. For instance, statins have shown a promising role in improving endothelial function and reducing oxidative stress by increasing the production of NO17. Based on the above, we suggest that patients with demonstrable PR on CTCA should be commenced on high-intensity statins.
Aspirin therapy in patients with non-obstructive CAD is beneficial when they are at higher risk, including older age and the presence of other comorbidities such as hypertension and diabetes mellitus. Aspirin is a reasonable primary prevention strategy in patients with cardiovascular risk and with evidence of coronary atherosclerosis, including features of high-risk plaque20. However, the use of aspirin does confer a bleeding risk, and the overall benefit of aspirin needs to be weighed between the bleeding risk and cardiovascular preventive effect, especially in patients with low risk20. Our results could be applied to the selection of patients for aspirin therapy to prevent unwarranted aspirin prescription to reduce bleeding risk.
Predilection of PR in the left anterior descending artery (LAD)
We found that PR occurred mainly in the LAD (46%), and this finding is corroborated by histopathological studies of 2,964 hearts, which showed increased atherosclerosis preponderance of the left coronary artery, especially of the LAD, when compared with the right coronary system. Various angiographic, intravascular, and CT studies have also confirmed this finding2122.
The exact mechanism causing the predilection of PR in the LAD remains unclear. PR is thought to be the consequence of altered local haemodynamics due to increased shear stress at sites of luminal narrowing in response to plaque growth. Coronary artery haemodynamic studies have hypothesised that differences in shear stress, wall stress and phasic flow between the LAD and right coronary artery (RCA) may explain the susceptibility of the LAD to atherosclerosis and PR23.
Physiologically, the flow in the RCA is more uniform during the cardiac cycle. The oscillatory shear stress is less prominent in the susceptible regions of remodelling of the RCA compared to those of the left coronary artery24. The augmented oscillatory shear and wall stresses in the LAD – which experiences a significant systolic decline accompanied by a remarkable diastolic increment – during the cardiac cycle could explain its increased susceptibility to PR25. Additionally, several anatomical differences between the right and left coronary system appear to play a key role in regulating the local atherogenic environment, including the integrity of the endothelium and NO bioavailability26.
ACS events and need for revascularisation therapy
Surmely et al demonstrated that PR predominantly occurred in plaques of patients with ACS27. Similarly, Pflederer and his colleagues reported a higher incidence of positively remodelled culprit lesions in patients with ACS, compared to those with stable CAD28. A further study found that PR is the most frequent high-risk plaque feature, compared to spotty calcification and low attenuation plaque, in ACS patients29. These findings corroborated our outcome data, as we observed that patients with PR were more likely to be readmitted with ACS and undergo revascularisation therapy with either PCI or CABG. Our data were also comparable to earlier studies. Otsuka et al found that the ACS event rate in patients with vulnerable plaque features was 3.2%, which was similar to the 2.5% we noted in our patients with PR plaque30.
Limitations
There are several limitations to our study. Firstly, our data originated from a single centre hence, the distribution of our catchment population and departmental protocols might limit the generalisability of our results. Secondly, the images were manually interpreted by observers, which could introduce a degree of observer bias and variability. However, operators who assessed the CTCA images were level 2 CTCA-trained and had at least 1 year of experience in reporting CTCA images. Thirdly, we had a relatively small sample size, which could reduce the statistical power of our data and inflate our effect size. Nonetheless, our findings corroborated with numerous published studies. Finally, our exclusion criteria of patients with minimal CAD might introduce a degree of systematic selection bias.
Conclusions
Positive remodelling of coronary arteries on CTCA is associated with older age groups and male gender. In these patients, high-intensity statin and aspirin therapies may be beneficial in modulating positive remodelling, hence reducing the incidence of ACS and the need for revascularisation therapy. A larger, multicentre prospective study would be useful to confirm our findings.
Impact on daily practice
Detection of positive remodelling on CTCA is a reliable prognostic indicator of future cardiovascular events and, thus, presents a valuable opportunity for the initiation of primary prevention therapy including high-intensity statin and aspirin.
Acknowledgements
We would like to acknowledge Ruhan Munasinghe and Philip Spencer for their help in data collection and guidance on CTCA protocol respectively.
Conflict of interest statement
The authors have no conflicts of interest to declare.
