Shuvanan Ray, MD, DM; Priyam Mukherjee, MD, DM; Siddhartha Bandyopadhyay, MD, DM; Suman Karmakar, MD, DM; Sabyasachi Mitra, MBBS; Prithwiraj Bhattacharjee*, MD, DM
AsiaIntervention 2019;5:41-51, DOI: 10.4244/AIJ-D-18-00017
Department of Cardiac Intervention, Fortis Hospital Anandapur, Kolkata, India
Abstract
Aims: Although provisional stenting with a single drug-eluting stent has proven clinical efficacy in the treatment of bifurcation stenosis, some patients may require two stents. We propose a novel technique, called “nano-crush”, which is easy to perform and can be used in all bifurcation angles.
Methods and results: The feasibility of the nano-crush technique was confirmed in an in vitro bench test and intravascular ultrasound (IVUS) study. Subsequently, 42 patients with de novo coronary bifurcation stenosis were treated by this novel procedure using drug-eluting stents at our centre between January 2008 and December 2015. We experienced procedural success in all (100%) patients without any complications. The primary efficacy endpoint of the one-year incidence of major adverse cardiac events (MACE) was noted in three (7.14%) patients, comprising one case of cardiac death at nine months post procedure and two cases of repeat revascularisation due to in-stent restenosis. There were no cases of periprocedural myocardial infarction or stent thrombosis. Angiographic follow-up at one year indicated intact stent patency in the remaining patients.
Conclusions: Initial experience with the nano-crush technique demonstrates that it can be performed easily without any procedural complications. Further, the angiographic and clinical follow-up indicates that the nano-crush technique is associated with acceptable clinical outcomes in a real-world scenario.
Abbreviations
ACC: American College of Cardiology
AHA: American Heart Association
ARC: Academic Research Consortium
CRF: chronic renal failure
DK: double-kissing
IVUS: intravascular ultrasound
LAD: left anterior descending artery
LCL: lower confidence limit
LCx: left circumflex artery
LMCA: left main coronary artery
MACE: major adverse cardiac events
NC: non-compliant
OCT: optical coherence tomography
POT: proximal optimisation technique
TIMI: Thrombolysis In Myocardial Infarction
UCL: upper confidence limit
Introduction
In unselected coronary bifurcation lesions, the provisional stenting technique with a single drug-eluting stent in the main branch has proven clinical efficacy1-3. However, some patients will still require two stents for the proper treatment of their bifurcation stenosis. Various elective double-stenting techniques are available; some of these double-stenting strategies have undergone various modifications in order to make them simpler and to optimise clinical outcomes. The “culotte” technique is one such long-standing strategy for the management of bifurcation lesions. Its efficacy has been documented in various trials (NORDIC I and BBC ONE)4,5, but has been questioned for distal left main bifurcation treatment6,7. Similarly, the “crush” technique has evolved over time after its first description by Colombo et al8. Although the short-term outcomes with the crush technique were encouraging, the midterm or long-term outcomes remain not fully satisfactory due to the high risk of periprocedural stent thrombosis and subsequent restenosis. To overcome these drawbacks, the “double-kissing (DK) crush” technique9, and “mini-crush” technique10 were developed. Their clinical performance has been appraised in studies. Further modifications of the crush technique have also been documented11.
In general, experts believe that an ideal crush technique should: (a) have a limited length of crushed segment in the main branch; (b) use an NC balloon in the main branch to crush the side branch stent; (c) push away the first layer of stent struts from the side branch orifice by performing a first kissing balloon inflation after stent crush to appose the struts fully on the carina side, thus increasing the success of final kissing balloon inflation8. Against this background, we have developed a novel technique considering all three points which are necessary for an optimum outcome in an elective double-stent technique for the treatment of bifurcation lesions. The proposed technique is easy to perform and can be used in almost all bifurcation angles. We have called our technique “nano-crush”. A detailed description of the technique and clinical experience is reported here.
Methods
STUDY POPULATION
A retrospective observational single-centre study was conducted at our centre. Patients with de novo coronary bifurcation lesions, who were treated with the nano-crush stenting technique between January 2008 and December 2015, were analysed. Here, patients were treated with the nano-crush technique if they had symptomatic de novo coronary bifurcation lesion stenosis with (a) diameter narrowing ≥50% in both main branch and side branch, (b) Medina class 1,1,1, and (c) vessel size ≥2.75 mm in the main branch and ≥2.5 mm in the side branch by visual estimation on coronary angiography. Exclusion criteria were: (a) contraindications to prolonged use of dual antiplatelet drugs; (b) life expectancy <1 year; (c) left ventricular ejection fraction <30%; and (d) acute myocardial infarction and acute coronary syndrome patients before stabilisation. All patients provided informed consent for the procedure and subsequent data collection and analysis for research purposes.
NANO-CRUSH TECHNIQUE
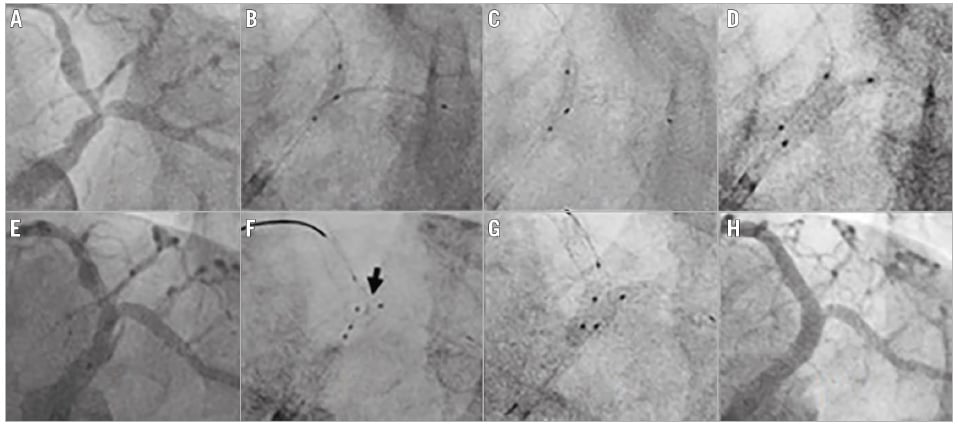
For significant coronary bifurcation lesions (Figure 1A), the nanocrush stenting technique can be performed with a 6 Fr guide catheter, but a 7 Fr guide catheter is preferable. Proper dilatation of both the branches, main branch and side branch, is performed. In calcified vessels, plaque modification may be needed with a scoring balloon, cutting balloon or rotational atherectomy. After vessel preparation, a stent is placed in the side branch according to the distal diameter and a non-compliant balloon (one size smaller than the distal diameter of the artery, e.g., 2.5 mm balloon in 3.5 mm artery) is placed in the main branch (Figure 1B). Then, the main branch balloon is inflated at nominal pressure and the side branch stent is pulled to the inflated balloon (so that some part of the side branch stent enters into the main branch) and the side branch stent is deployed at nominal pressure. Both balloons are deflated and the side branch balloon is pulled into the main branch so that 50% of it remains inside the side branch stent and is dilated at high pressure to open the ostium of the side branch nicely. Subsequently, the side branch balloon is deflated and the main branch balloon is inflated at a high pressure (≥20 atm) to crush the edge of the side branch stent in the main branch (Figure 1C). First kissing balloon inflation with the side branch stent balloon (pulled inside the main branch) and the main branch balloon is carried out at 14-16 atm (Figure 1D). The side branch balloon and the wire along with the main branch balloon are removed. Then, the main branch stent is chosen according to the distal diameter of the vessel and the stent is placed and deployed at nominal pressure (Figure 1E). The proximal optimisation technique (POT) is carried out with a 0.5 mm larger NC balloon in the proximal part of the main branch stent and the side branch is accessed trans-strut through the middle struts of the main branch stent overhanging the side branch ostium by a second wire (Figure 1F). The final kissing balloon inflation is performed with appropriately sized non-compliant balloons at a moderate 14-16 atm (Figure 1G). Post-procedural check angiography is performed to evaluate the final result (Figure 1H).
IN VITRO BENCH TESTING AND IVUS STUDIES
Prior to evaluation in real-world patients, the feasibility of this novel procedure was evaluated using an in vitro bench test and IVUS study. The in vitro bench test was conducted using a bifurcation model by fusing 3.0 mm and 2.5 mm thermoplastic tubes in Y fashion according to Finet’s model based on fractal arguments. The deployment of stents was carried out in the model in a water bath with all steps such as side branch wire access, stent deployment in the side branch (Figure 2A), crushing and first kissing balloon inflation (Figure 2B), stent deployment in the main branch (Figure 2C), final kissing balloon (Figure 2D), and final result (Figure 2E), and were viewed live and recorded fluoroscopically. Intravascular ultrasound (Atlantis™ SR Pro; Boston Scientific, Marlborough, MA, USA) was also performed in this model along with in vitro IVUS studies.
INTERVENTIONAL PROCEDURE IN REAL-WORLD PATIENTS
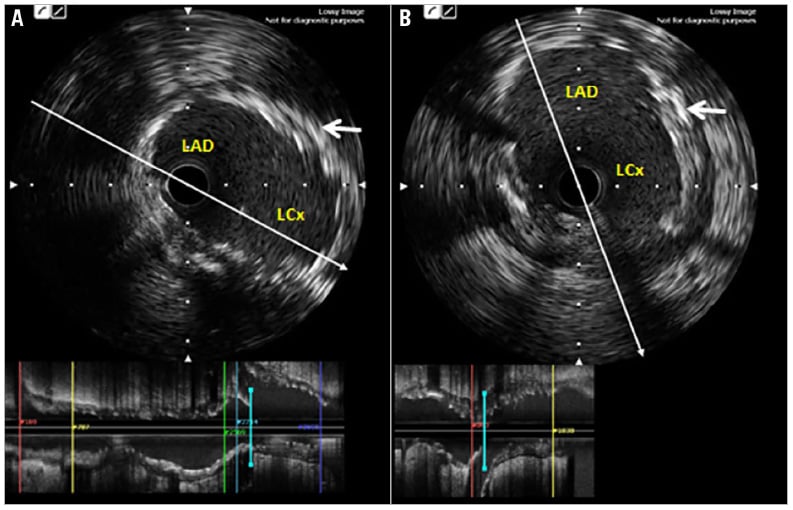
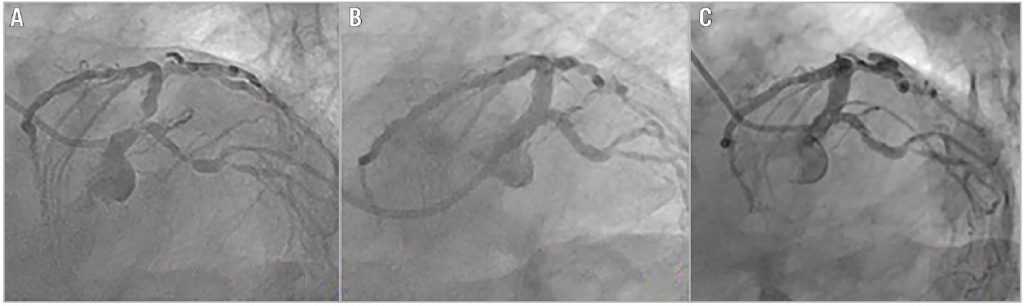
We used the femoral access in all cases. All patients were treated with dual antiplatelet therapy (DAPT) before the procedure, according to ACC/AHA guidelines12 (e.g., aspirin and either clopidogrel, prasugrel or ticagrelor after a proper loading dose). During the intervention, heparin was given intra-arterially as anticoagulant to maintain an activated coagulation time >250 seconds. The nano-crush technique was performed as detailed above. IVUS was used in cases with a significant bifurcation lesion in the left main coronary artery (Figure 3A, Figure 3B) and in cases which required rotational atherectomy. After the procedure, all patients were advised to maintain DAPT (i.e., aspirin and clopidogrel or prasugrel or ticagrelor) for at least 12 months.
DATA COLLECTION AND PATIENT FOLLOW-UP
Demographic data including age, gender, cardiovascular risk factors, medical history, and clinical presentation were collected from hospital records. Details of the affected lesions and implanted stents were obtained from angiography and angioplasty reports. Adverse events were monitored during hospital stay. Subsequently, patients were called into the out-patient department after 1 month, 3 months, 6 months and 12 months for clinical evaluation. A mandatory coronary angiogram was planned after one year in all cases.
STUDY ENDPOINTS
The procedural success rate was considered to be the performance endpoint. The primary efficacy endpoint was the one-year incidence of major adverse cardiac events (MACE). Events of Academic Research Consortium (ARC)-defined stent thrombosis were also considered to be an additional safety endpoint12.
DEFINITIONS AND CLINICAL ENDPOINTS
Procedural success was defined as successful delivery and deployment of the coronary stents at the intended coronary bifurcation lesions and successful withdrawal of the stent delivery systems with achievement of a final diameter stenosis of <30% and TIMI grade 3 flow in both the main branch and side branch vessels, without the occurrence of death, MI, and repeat revascularisation of the treated lesions in the index hospitalisation. MACE was defined as a composite of cardiac death, myocardial infarction, and any repeat revascularisation in the target vessel. All deaths were considered cardiac in origin unless otherwise documented.
Myocardial infarction was defined as either development of new ischaemic ST-T changes or pathological Q-waves in at least two contiguous leads of the electrocardiogram or elevation of cardiac troponin >5 times the normal13. Target vessel revascularisation was defined as any repeat revascularisation (either percutaneous or surgical) procedure in the target vessel. Renal failure was defined as an estimated glomerular filtration rate (eGFR) <45 mL/min/1.73 m2. Stent thrombosis was defined using the Academic Research Consortium (ARC) definitions14. The stent thrombosis was considered as “definite” when it was detected angiographically, “probable” if the patient had a target vesselrelated myocardial infarction or died of a coronary event within the first 30 days, and “possible” if any unexplained death occurred from 30 days after the index procedure until the final follow-up.
STATISTICAL ANALYSIS
All categorical variables were expressed as counts (percentage, %) with number of patients (n=42) as denominator; continuous variables were expressed as mean±standard deviation (SD). Cox regression analysis was used to find the hazard ratio and identify predictors for the occurrence of MACE. The time-dependent occurrence of MACE was obtained using the Kaplan-Meier method with log-rank comparison. All tests were two-sided and a p-value less than 0.05 was considered statistically significant. Statistical software R was used for analysis (with the package “survival”), and the R statistical function used coxph and survfit (R Foundation for Statistical Computing, Vienna, Austria).
Figure 1. Angiography showing the steps of the nano-crush technique. A) Vessel anatomy showing bifurcation lesion in the distal LM and the ostio-proximal LAD and LCx. B) Side branch stent positioning with an inflated NC balloon in the main branch. C) Balloon crushing of the side branch stent. D) First kissing balloon inflation with the stent balloon and an NC balloon. E) Positioning of the main branch stent after removing the balloon and stent from the side branch. F) Positioning NC balloons in the main branch and side branch after re-crossing the side branch. Arrow shows minimally crushed stent at the carina. G) Final kissing balloon inflation. H) Final result.
Figure 2. Nano-crush bench test. A) The main branch balloon is inflated at nominal pressure and the side branch stent is pulled to the inflated balloon. B) First kissing balloon inflation with the side branch stent balloon (pulled inside the main branch) and main branch balloon carried out at 14-16 atm. C) The main branch stent is placed and deployed at nominal pressure. D) Final kissing balloon inflation is performed. E) Final result showing minimally crushed stent.
Figure 3. IVUS images of a patient with LMCA bifurcation stenting using the nano-crush technique. A) LAD to LMCA pullback showing full coverage of the LCx ostium by the stent strut. B) LCx to LMCA pullback. White arrow indicates minimally crushed stent segment in panels A and B.
Results
IN VITRO BENCH TESTING
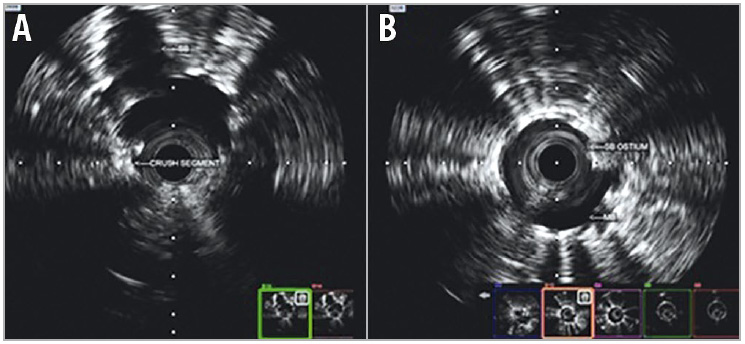
The in vitro bench test and in vitro IVUS studies showed proper covering of the carina and circular opening of the side branch stent, without any major distortion of the side branch stent at the ostium (Figure 4).
BASELINE DEMOGRAPHICS
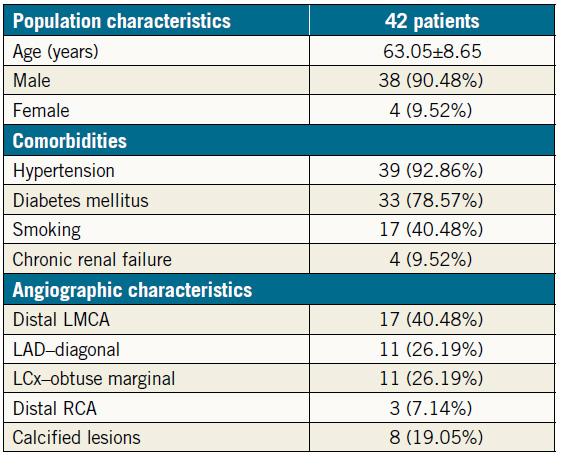
Baseline clinical characteristics for the 42 patients analysed in the study are shown in Table 1. In brief, the mean age of the study population was 63.05±8.65 years; 38 (90.48%) were male. Hypertension was present in 39 (92.86%) patients, while type 2 diabetes mellitus was present in 33 (78.57%) patients. Four (9.52%) patients had chronic renal failure (CRF) and were maintained on haemodialysis. In addition, 17 (40.48%) patients had left main coronary artery (LMCA) bifurcation disease. Eight (19.05%) patients with calcified arteries required rotational atherectomy for plaque modification. Four (9.52%) patients with an LMCA bifurcation required rotational atherectomy.
Figure 4. IVUS pullback. A) Main branch. B) Side branch to main branch.
Table 1. Population characteristics (n=42).
CLINICAL OUTCOMES
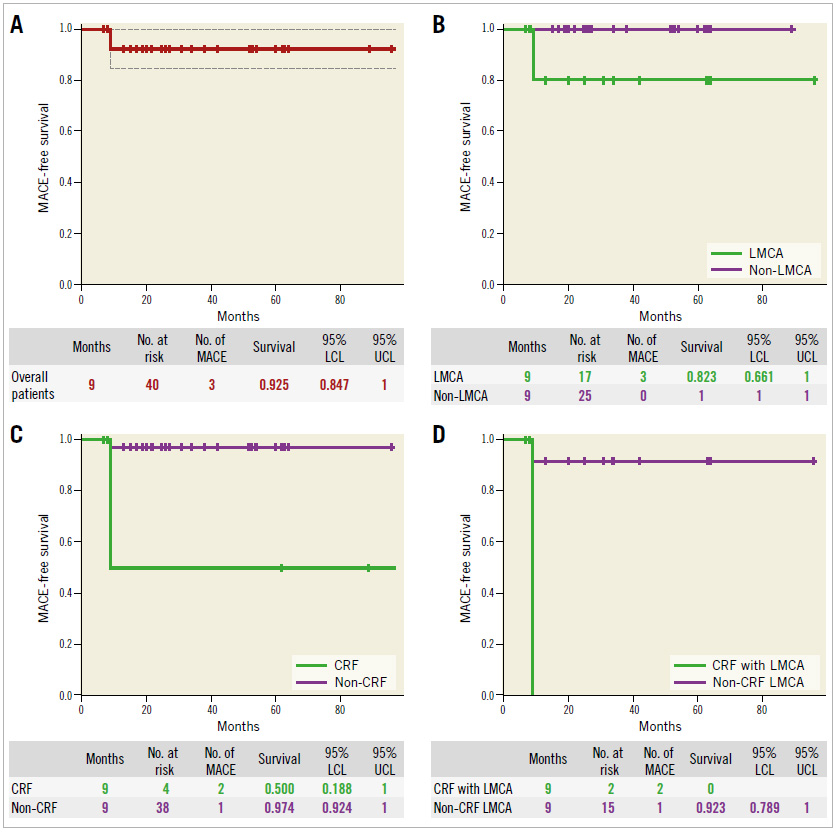
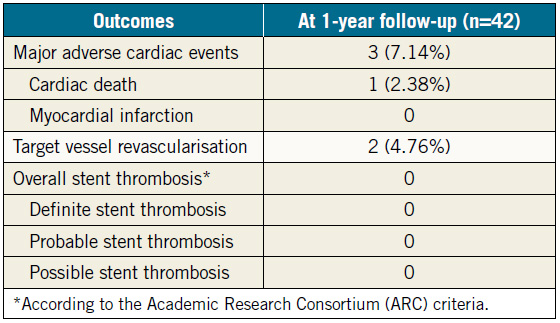
The procedural success rate was 100% with no cases of periprocedural myocardial infarction or other complications. All patients were followed up for one year and underwent follow-up angiography. The median follow-up period was 24 months (IQR: 16-53 months). Subsequently, the primary endpoint of cumulative MACE at one-year follow-up was noted in three (7.14%; 95% confidence interval [CI]: 0-14.93) patients, which included one case of cardiac death due to left ventricular failure nine months after the index procedure and two cases of repeat revascularisation due to diffuse in-stent restenosis after nine months. Angiographic follow-up in the remaining patients indicated intact stent patency at one year (Figure 5). There was no stent thrombosis event in any patient up to one-year follow-up. The detailed study outcomes are shown in Table 2. The Kaplan- Meier MACE-free survival curve for the overall patient population is illustrated in Figure 6A.
PREDICTORS OF ADVERSE EVENTS AFTER THE NANOCRUSH TECHNIQUE
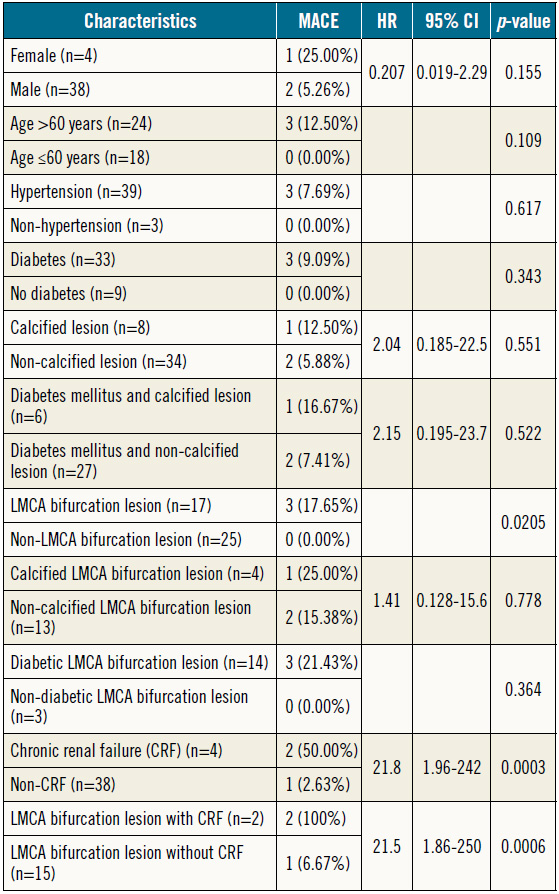
The Cox regression analysis findings for the predictors of adverse events after the nano-crush technique are given in Table 3. No significant difference was reported in the occurrence of MACE with respect to gender (male vs. female), age (≤60 years vs. >60 years), type 2 diabetes mellitus, hypertension, and calcified lesion. However, a significant difference in MACE was noted with respect to LMCA lesions (17.65% vs. 0.0%, p=0.0205) (Figure 6B) and with respect to chronic renal failure (50.00% vs. 2.63%; HR 21.8, p=0.0003) (Figure 6C). Further, a significant difference in the number of MACE was also noted for patients having LMCA lesions and CRF vs. patients having LMCA lesions and no CRF (100% vs. 7.69%; HR 21.5, p=0.0006) (Figure 6D).
Figure 5. Coronary angiography of a patient showing significant bifurcation lesion at baseline (A), good coronary flow after stenting with the
nano-crush technique (B), and stent patency at one-year follow-up (C).
Figure 6. Kaplan-Meier MACE-free survival curve. A) Overall patient population. B) Patients with an LMCA bifurcation lesion vs. patients
with non-LMCA bifurcation lesions. C) Patients with CRF vs. patients without CRF. D) Patients with an LMCA bifurcation lesion with CRF
vs. patients with LMCA bifurcation lesions without CRF.
Table 2. Clinical outcomes at 1-year follow-up (n=42).
Table 3. Evaluation of predictors of MACE after nano-crush technique.
Discussion
Bifurcations vary not only in terms of anatomy (plaque burden, location of the plaque, angle between the branches, diameter of the branches, and bifurcation site), but also in relation to dynamic changes in the anatomy during treatment (plaque shift, dissection). As a result, no two bifurcations are identical and no single strategy exists that can be applied to every bifurcation15.
A provisional single-stent strategy is currently regarded as the default strategy for the treatment of bifurcation lesions. However, this approach cannot be applied broadly across all bifurcation lesions because of the likelihood of side branch compromise in the presence of high-risk features (e.g., significant side branch ostial disease), or because the side branch has significant disease beyond its ostium requiring treatment. In such circumstances, a two-stent strategy is required in a bid to maintain optimal vessel patency and blood flow in both the main and side branches15.
An ideal two-stent strategy should offer: (a) optimal coverage of the side branch ostium, with no gaps between the main branch and the side branch stents; (b) minimal distortion of the side branch stent at the ostium; (c) minimal overlap between the main branch stent and the side branch stent; (d) short procedural time; (e) minimal requirement of additional guidewires and balloons; (f) the ability to maintain control of both the main branch and the side branch, so that there is little risk of compromised blood flow in either branch; (g) an easy to perform, reproducible, and predictable method16-19. Considering these, a number of different two-stent techniques have been described in an attempt to optimise the immediate and long-term results of the side branch following treatment. To date, no single approach is able to deliver all the desired attributes as described and each of these strategies has its own limitations, including stent distortion, inadequate ostial coverage and multiple stent layers that contribute to restenosis20,21.
The main disadvantage of T-stenting is the risk of missing the side branch ostium. Burzotta et al described modified T-stenting by intentionally protruding the side branch stent within the main branch stent22. Though this technique ensures that the side branch ostium is not missed, there is a risk of the side branch stent inadvertently protruding too much inside the main branch. In addition, access to the main branch stent struts with balloons and stents could be difficult in some cases such as a tortuous side branch and a long side branch stent.
The crush technique was the first of the elective double stenting techniques introduced to ensure ostial side branch coverage, which was one of the limitations in the classic “T-stent” technique8. Though the crush technique yielded comparatively better clinical outcomes than the T-stent technique23, there were several concerns about the original crush technique. One such concern involves the 3-4 mm of drug-eluting stent overlap at the bifurcation. A study on long-term (nine months) follow-up with the crush technique for coronary bifurcation lesions has demonstrated a stent thrombosis rate of up to 4.3%, while the restenosis rate, mostly at the side branch ostium, was as high as 25.3%24.
Later, the importance of appropriate final kissing balloon inflation with this technique was established25. The use of mandatory high-pressure dilatation of the side branch and kissing balloon inflation during the crush technique displayed a reduced restenosis rate of 8.6% in a more recent multicentre registry. However, the rate of stent thrombosis with this technique remained unchanged at 3.3% despite the kissing inflation23. The subsequently developed mini-crush technique (with step balloon crushing)26, which calls for a reduced stent overlap of 1-2 mm, demonstrated improved final kissing inflation rates, low MACE rates, and a very low restenosis rate of 2.0% at the side branch ostium in a small patient cohort10.
Although the DK crush technique satisfies many of the ideal features for a two-stent strategy, it is complex and needs crossing of a crushed segment through the proximal struts, which is timeconsuming, requires more than one balloon, and is often difficult19. It should also be noted that the DK crush technique is essentially a “mini double-kissing crush” technique, where the side branch is stented with a 1-2 mm protrusion inside the main branch. In acute side branch angles (Y-shaped), the side branch ostium is longer, and oval-shaped. Such an anatomic configuration implies the need for wider protrusion of the side branch stent inside the main, resulting in a longer neocarina19,27. Further, the bifurcation angle (>50 degrees) has been noted to be an independent predictor of MACE after crush stenting24. This may be due to inadequate expansion of the side branch stent ostium, even after kissing inflation. Considering these, striving to limit protrusion while implanting the side branch stent becomes vital19.
At our centre, extensive experience of treating bifurcation stenosis with “crush” stenting helped us make some refinements to it. These include: (a) limiting the length of the crushed stent segment (mini/micro/nano) during stent implantation in the side branch; (b) use of a balloon in the main branch to crush the side branch stent (step crush) in place of a stent as in classic crush techniques; and (c) performing first kissing balloon inflation after crush in order to push away the first layer of stent struts from the side branch orifice and to appose the stents fully on the carina side (securing wire re-crossing towards the side branch), thus increasing the rate and success of final kissing balloon inflation (DK crush).
The nano-crush technique proposed by us is much simpler and easy to perform. The technique is compatible with a 6 Fr guide catheter. In addition, the wire in the side branch is never lost before adequate deployment and crushing, and further optimises opening and apposition by the first kissing balloon inflation. Our method also overcomes the limitations of the DK crush technique as the one size smaller balloon in the main branch is inflated at nominal pressure (i.e., 3.0 mm balloon in a 3.5 mm vessel), which ensures some amount of stent strut protrusion into the main branch, and prevents the side branch stent protruding into the main branch excessively, while covering the ostium of the side branch completely. In high-angled (≥70˚) bifurcation lesions, the edges, medial and lateral parts of the side branch stent struts will protrude into the main branch. On the other hand, in patients with a narrow angle bifurcation (≤70°), the operator should be careful to ensure that there is minimal entry of the side branch stent into the main branch before deployment.
Unlike the modified T-stenting technique, there will be definite crush in this technique, though minimal, and that is why we call this technique nano-crush. The guidewire re-crosses easily through the expanded side branch ostium due to a lesser metal load and the first kissing balloon inflation. Further, the minimal amount of metal which may remain in the medial wall of the side branch may form a metallic neocarina after the final kissing balloon, indicating that the side branch ostium is not fully covered by the crushed segment and the retained stent balloon comes out without much difficulty. The same observation was confirmed in the bench test and in vitro IVUS studies before performing the technique in real-world patients. We did not experience any difficulty in removing the side branch balloon after crushing with the main branch balloon in any of our cases. Here, the trick is to ensure minimal protrusion of the side branch stent by selecting the correct size of main branch balloon. Our initial experience of managing patients with bifurcation lesions with the nanocrush technique suggested good immediate and long-term clinical results. Further, the technique is easy to perform and can be used in all bifurcation angles.
One of the anticipated pitfalls of this technique is a short proximal segment before bifurcation. In that case it would be difficult to wire the side branch after the stent placement in the main branch. Hence, it is advisable to choose stents with minimal or no balloon overhang for accurate positioning of the side branch stent. It should also be noted that, if the operator recrosses the guidewire in the expanded side branch stent ostium and not in the crushed lateral cell, the stenting would be only a modification of the modified T-stenting technique. During the first kissing balloon inflation, the side branch stent strut near the carina may open towards the distal main branch or towards the side branch ostium, depending upon the angle of bifurcation. If it opens towards the distal main branch, the wire will re-cross through the middle strut of the main branch stent. If, due to a narrow angle, the side branch stent protrudes more inside the main branch near the carina, the struts are more likely to be displaced towards the side branch ostium during the first kissing balloon inflation. Due to the relatively lower metal load compared to the classic crush, re-entry will be relatively easy through the middle strut. Nevertheless, rewiring of the side branch through the distal strut should be avoided. Subsequently, high-pressure final kissing balloon inflation is recommended in all cases. IVUS could be helpful in identifying suboptimal opening of the stent struts at the side branch ostium.
In our study, we observed significantly higher MACE rates in patients with bifurcation lesions in the LMCA as compared to patients with bifurcation lesions in other vessels. The COBIS II registry demonstrated similar outcomes, i.e., that two-stent bifurcation techniques in LMCA bifurcation lesions are associated with more target vessel failure as compared to non-LMCA bifurcation lesions28. In our study, we also observed significantly higher MACE rates in CRF patients with bifurcation lesions as compared to non-CRF patients with bifurcation lesions. Further, the coexistence of CRF with an LMCA bifurcation lesion was also associated with significantly higher MACE rates. In line with these findings, a recent study by Cho et al also demonstrated that CRF is an independent predictor of MACE in LMCA bifurcation stenting29.
Overall, our study shows that the nano-crush is an optimum elective double stent technique for bifurcation lesions which is safe and can be performed easily, does not require complex wiring and ballooning, can be performed with less hardware and is 6 Fr guide catheter-compatible. The bench test and in vitro IVUS examination did not show any major distortion of the side branch stent at the ostium and this is reflected in the actual clinical outcome. This operator-friendly and safe technique can be adopted as a two-stent bifurcation strategy in a real-world scenario.
Study limitations
The major limitations of our study include its retrospective observational study design and the small number of patients. The lack of comparison of clinical and angiographic outcomes with the nanocrush technique vs. other strategies could also be considered a limitation of the study. In addition, although angiographic follow-up was mandatory in all patients at one-year follow-up, we did not perform IVUS, or optical coherence tomography (OCT) imaging in all patients. In addition, acute myocardial infarction and thrombotic lesions were excluded from the study.
Conclusions
These results demonstrate that the nano-crush technique is associated with acceptable clinical outcomes with no episodes of definite or probable stent thrombosis. Further studies should compare the nano-crush technique with other double-stent strategies in the management of bifurcation lesions.
Impact on daily practice
Management of coronary bifurcation lesions with a dedicated two-stent technique is technically challenging. We propose a novel technique, called nano-crush, which can be performed easily and can be used in all bifurcation angles. In this technique, minimal protrusion of the proximal part of the side branch stent was controlled by main branch dilation with a small size balloon. This novel technique has the following advantages: (a) minimal metal overlap; (b) less metallic carina formation compared to other crush stenting techniques; and (c) less geographic miss in the side branch ostium compared to T-stenting. The feasibility of this technique has been confirmed in bench testing. In addition, an acceptably lower MACE (7.14%) rate at long-term follow-up was observed in our initial experience in a real-world setting in patients with true bifurcation lesions with large side branches (>2.5 mm).
Acknowledgements
The authors gratefully acknowledge the contributions of Mr Dipankar Sadhukhan, Mr Sujoy Bhattacharjee, Mr Niladri Ghora, Mrs Susweta Banerjee, Ms Arpita Nath and all other cath lab staff and nurses of the Fortis Hospital Anandapur, Kolkata, India.
Conflict of interest statement
The authors have no conflicts of interest to declare.
References
1. Louvard Y, Thomas M, Dzavik V, Hildick-Smith D, Galassi AR, Pan M, Burzotta F, Zelizko M, Dudek D, Ludman P, Sheiban I, Lassen JF, Darremont O, Kastrati A, Ludwig J, Iakovou I, Brunel P, Lansky A, Meerkin D, Legrand V, Medina A, Lefèvre T. Classification of coronary artery bifurcation lesions and treatments: time for a consensus! Catheter Cardiovasc Interv. 2008;71:175-83.
2. Fernec M, Gick M, Keinzle RP, Bestehorn HP, Werner KD, Comberg T, Kuebler P, Büttner HJ, Neumann FJ. Randomized trial on routine versus provisional T-stenting in the treatment of de novo coronary bifurcation lesions. Eur Heart J. 2008;29:2859-67.
3. Brend P, Lefevre T, Darremont O, Louvard Y. Provisional T-stenting and kissing balloon in the treatment of coronary bifurcation lesions: results of French multicenter “TULIPE” study. Catheter Cardiovasc Interv. 2006;68:67-73.
4. Steigen TK, Maeng M, Wiseth R, Erglis A, Kumsars I, Narbute I, Gunnes P, Mannsverk J, Meyerdierks O, Rotevatn S, Niemelä M, Kervinen K, Jensen JS, Galløe A, Nikus K, Vikman S, Ravkilde J, James S, Aarøe J, Ylitalo A, Helqvist S, Sjögren I, Thayssen P, Virtanen K, Puhakka M, Airaksinen J, Lassen JF, Thuesen L; Nordic PCI Study Group. Randomized study on simple versus complex stenting of coronary artery bifurcation lesions: the Nordic bifurcation study. Circulation. 2006;114:1955-61.
5. Hildick-Smith D, de Belder AJ, Cooter N, Curzen NP, Clayton TC, Oldroyd KG, Bennett L, Holmberg S, Cotton JM, Glennon PE, Thomas MR, Maccarthy PA, Baumbach A, Mulvihill NT, Henderson RA, Redwood SR, Starkey IR, Stables RH. Randomized trial of simple versus complex drug-eluting stenting for bifurcation lesions: the British Bifurcation Coronary Study: old, new and evolving strategies. Circulation. 2010;121:1235-43.
6. Chen SL, Xu B, Han YL, Sheiban I, Zhang JJ, Ye F, Kwan TW, Paiboon C, Zhou YJ, Lv SZ, Dangas GD, Xu YW, Wen SY, Hong L, Zhang RY, Wang HC, Jiang TM, Wang Y, Sansoto T, Chen F, Yuan ZY, Li WM, Leon MB. Clinical Outcome After DK Crush Versus Culotte Stenting of Distal Left Main Bifurcation Lesions: The 3-Year Follow-Up Results of the DKCRUSH-III Study. JACC Cardiovasc Interv. 2015;8:1335-42.
7. Chevalier B, Glatt B, Royer T, Guyon P. Placement of coronary stents in bifurcation lesions by the culotte technique. Am J Cardiol. 1998;82:943-9.
8. Colombo A, Stankovic G, Orlic D, Corvaja N, Liistro F, Airoldi F, Chieffo A, Spanos V, Montorfano M, Di Mario C. Modified T-stenting with crushing for bifurcation lesions: immediate results and 30-day outcome. Catheter Cardiovasc Interv. 2003;60:145-51.
9. Zhang JJ, Chen SL. Classic crush and DK crush stenting techniques. EuroIntervention. 2015;11:V102-5.
10. Galassi AR, Colombo A, Buchbinder M, Grasso C, Tomasello SD, Ussia GP, Tamburino C. Long-term outcomes of bifurcation lesions after implantation of drug-eluting stents with the “minicrush technique”. Catheter Cardiovasc Interv. 2007;69:976-83.
11. Yang HM, Tahk SJ, Woo SI, Lim HS, Choi BJ, Choi SY, Yoon MH, Park JS, Zheng M, Hwang GS, Kang SJ, Shin JH. Longterm clinical and angiographic outcomes after implantation of sirolimus-eluting stents with a “modified mini-crush” technique in coronary bifurcation lesions. Catheter Cardiovasc Interv. 2009;74: 76-84.
12. Levine GN, Bates ER, Blankenship JC, Bailey SR, Bittl JA, Cercek B, Chambers CE, Ellis SG, Guyton RA, Hollenberg SM, Khot UN, Lange RA, Mauri L, Mehran R, Moussa ID, Mukherjee D, Nallamothu BK, Ting HH; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines; Society for Cardiovascular Angiography and Interventions. 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. Catheter Cardiovasc Interv. 2013;82: E266-355.
13. Thygesen K, Alpert JS, Jaffe AS, Simoons ML, Chaitman BR, White HD; Joint ESC/ACCF/AHA/WHF Task Force for the Universal Definition of Myocardial Infarction, Katus HA, Lindahl B, Morrow DA, Clemmensen PM, Johanson P, Hod H, Underwood R, Bax JJ, Bonow RO, Pinto F, Gibbons RJ, Fox KA, Atar D, Newby LK, Galvani M, Hamm CW, Uretsky BF, Steg PG, Wijns W, Bassand JP, Menasché P, Ravkilde J, Ohman EM, Antman EM, Wallentin LC, Armstrong PW, Simoons ML, Januzzi JL, Nieminen MS, Gheorghiade M, Filippatos G, Luepker RV, Fortmann SP, Rosamond WD, Levy D, Wood D, Smith SC, Hu D, Lopez-Sendon JL, Robertson RM, Weaver D, Tendera M, Bove AA, Parkhomenko AN, Vasilieva EJ, Mendis S. Third universal definition of myocardial infarction. Circulation. 2012;126:2020-35.
14. Cutlip DE, Windecker S, Mehran R, Boam A, Cohen DJ, van Es GA, Steg PG, Morel MA, Mauri L, Vranckx P, McFadden E, Lansky A, Hamon M, Krucoff MW, Serruys PW; Academic Research Consortium. Clinical end points in coronary stent trials: a case for standardized definitions. Circulation. 2007;115:2344-51.
15. Stankovic G, Mehmedbegovic Z, Zivkovic M. Bifurcation Coronary Lesions – Approaches To Bifurcation Management. Interventional Cardiology Review. 2010;5:53-57.
16. Colombo A, Ruparelia N. The Relentless Attempt to Perfect the 2-Stent Technique. JACC Cardiovasc Interv. 2015;8:960.
17. Lassen JF, Holm NR, Banning A, Burzotta F, Lefèvre T, Chieffo A, Hildick-Smith D, Louvard Y, Stankovic G. Percutaneous coronary intervention for coronary bifurcation disease: 11th consensus document from the European Bifurcation Club. EuroIntervention. 2016;12:38-46.
18. Latib A, Colombo A. Bifurcation disease: what do we know, what should we do? JACC Cardiovasc Interv. 2008;1:218-26.
19. Sawaya FJ, Lefèvre T, Chevalier B, Garot P, Hovasse T, Morice MC, Rab T, Louvard Y. Contemporary approach to coronary bifurcation lesion treatment. JACC Cardiovasc Interv. 2016; 9:1861-78.
20. Dou K, Zhang D, Xu B, Yang Y, Yin D, Qiao S, Wu Y, Yan H, You S, Wang Y, Wu Z, Gao R, Kirtane AJ. An angiographic tool for risk prediction of side branch occlusion in coronary bifurcation intervention; the RESOLVE score system (Risk prEdiction of Side branch OccLusion in coronary bifurcation interVEntion). JACC Cardiovasc Interv. 2015;8:39-46.
21. Moussa I, Costa RA, Leon MB, Lansky AJ, Lasic Z, Cristea E, Trubelja N, Carlier SG, Mehran R, Dangas GD, Weisz G, Kreps EM, Collins M, Stone GW, Moses JW. A prospective registry to evaluate sirolimus-eluting stents implanted at coronary bifurcation lesions using the “crush technique”. Am J Cardiol. 2006;97:1317-21.
22. Burzotta F, Gwon HC, Hahn JY, Romagnoli E, Choi JH, Trani C, Colombo A. Modified T-stenting with intentional protrusion of the side-branch stent within the main vessel stent to ensure ostial coverage and facilitate final kissing balloon: the T-stenting and small protrusion technique (TAP-stenting). Report of bench testing and first clinical Italian-Korean two-centre experience. Catheter Cardiovasc Interv. 2007;70:75-82.
23. Ge L, Iakovou I, Cosgrave J, Agostoni P, Airoldi F, Sangiorgi GM, Michev I, Chieffo A, Montorfano M, Carlino M, Corvaja N, Colombo A. Treatment of bifurcation lesions with two stents: one year angiographic and clinical follow up of crush versus T stenting. Heart. 2006;92:371-6.
24. Hoye A, Iakovou I, Ge L, van Mieghem CA, Ong AT, Cosgrave J, Sangiorgi GM, Airoldi F, Montorfano M, Michev I, Chieffo A, Carlino M, Corvaja N, Aoki J, Rodriguez Granillo GA, Valgimigli M, Sianos G, van der Giessen WJ, de Feyter PJ, van Domburg RT, Serruys PW, Colombo A. Long-term outcomes after stenting of bifurcation lesions with the “crush” technique: predictors of an adverse outcome. J Am Coll Cardiol. 2006;47:1949-58.
25. Ge L, Airoldi F, Iakovou I, Cosgrave J, Michev I, Sangiorgi GM, Montorfano M, Chieffo A, Carlino M, Corvaja N, Colombo A. Clinical and angiographic outcome after implantation of drug-eluting stents in bifurcation lesions with the crush stent technique: importance of final kissing balloon post-dilation. J Am Coll Cardiol. 2005;46:613-20.
26. Collins N, Dzavik V. A modified balloon crush approach improves side branch access and side branch stent apposition during crush stenting of coronary bifurcation lesions. Catheter Cardiovasc Interv. 2006;68:365-71.
27. Dzavik V, Kharbanda R, Ivanov J, Ing DJ, Bui S, Mackie K, Ramsamujh R, Barolet A, Schwartz L, Seidelin PH. Predictors of long-term outcome after crush stenting of coronary bifurcation lesions: importance of bifurcation angle. Am Heart J. 2006;152: 762-9.
28. Song YB, Hahn JY, Yang JH, Choi SH, Choi JH, Lee SH, Jeong MH, Kim HS, Lee JH, Yu CW, Rha SW, Jang Y, Yoon JH, Tahk SJ, Seung KB, Oh JH, Park JS, Gwon HC. Differential prognostic impact of treatment strategy among patients with left main versus non-left main bifurcation lesions undergoing percutaneous coronary intervention: results from the COBIS (Coronary Bifurcation Stenting) Registry II. JACC Cardiovasc Interv. 2014;7:255-63.
29. Cho S, Kang TS, Kim JS, Hong SJ, Shin DH, Ahn CM, Kim BK, Ko YG, Choi D, Song YB, Hahn JY, Choi SH, Gwon HC, Hong MK, Jang Y. Long-Term Clinical Outcomes and Optimal Stent Strategy in Left Main Coronary Bifurcation Stenting. JACC Cardiovasc Interv. 2018;11:1247-58.
To download, please click below.