Heart failure (HF) is a major global health crisis, affecting approximately 64 million individuals worldwide123. Persistent pulmonary venous congestion resulting from elevated left atrial pressure is a hallmark of HF, irrespective of left ventricular ejection fraction (LVEF)45. Notably, 50-60% of HF cases occur in patients with an LVEF >40%, classified as HF with preserved LVEF (HFpEF)678. Despite advancements in pharmacological and device-based therapies, HFpEF remains associated with high morbidity and mortality, often exceeding those of several cancers3. Currently, diuretics and sodium-glucose cotransporter-2 (SGLT2) inhibitors are the only two medications with guideline recommendations for HFpEF9.
Prolonged pulmonary venous congestion leads to pulmonary hypertension (PH), defined as a mean pulmonary arterial pressure (PAP) >20 mmHg and a pulmonary artery wedge pressure (PAWP) >15 mmHg at rest, as determined by right heart catheterisation (RHC)9. PH secondary to HF (PH-HF) is categorised into isolated postcapillary PH (IpcPH; pulmonary vascular resistance [PVR] ≤2 Wood units) and combined pre- and post-capillary PH (CpcPH; PVR >2 Wood units). The latter involves additional pulmonary vascular remodelling or vasoconstriction9. While targeted therapies for World Health Organization (WHO) Group I PH are not recommended for PH-HF1011, the presence of PH exacerbates HF severity. Given the pathophysiological roles of excessive vasoconstriction and sympathetic overactivation in HF1213, preclinical studies in canine and porcine models have demonstrated the safety and haemodynamic benefits of pulmonary artery denervation (PADN), including reductions in pulmonary arterial pressure and sympathetic activity. Subsequently1415, the PADN-5 study demonstrated that PADN significantly improved LVEF, cardiac output, and clinical outcomes in patients with CpcPH161718. However, the effects of PADN in HFpEF patients with IpcPH remain unknown. Furthermore, while guideline-directed medical therapy (GDMT) for HF with reduced ejection fraction (HFrEF) is well established, optimal pharmacological strategies for HFpEF require further study. This study aimed to evaluate the feasibility, safety, and efficacy of PADN in patients with HFpEF-induced IpcPH.
Methods
Study design and participants
This was a single-centre, proof-of-principle cohort study conducted in China. Eligible patients were ≥18 years old with chronic ischaemic or non-ischaemic cardiomyopathy (≥6 months duration) and stage C HF according to the ACC/AHA classification11. Patients had New York Heart Association (NYHA) Class III or ambulatory Class IV symptoms and had received GDMT for at least 3 months before enrolment, to maximally tolerated doses per ACC/AHA guidelines. Inclusion criteria included LVEF >40%, PAWP >15 mmHg, mean PAP >20 mmHg, and PVR ≤2 Wood units.
Exclusion criteria included indications for coronary revascularisation, recent stroke or thromboembolism (within 6 months), patent foramen ovale or atrial septal defect, atrial fibrillation requiring anticoagulation in patients with an intolerance to anticoagulant therapy, severe (grade 4) mitral regurgitation, or right atrial/right ventricular (RV) thrombus. Patients with a pacemaker implanted within the prior 3 months were also excluded.
PADN received regulatory approval for treating Group I PH from the National Medical Products Administration of China in December 2023 but remains unapproved for IpcPH. This study was conducted within a compassionate use framework for patients with severe or life-threatening conditions who had exhausted conventional therapies. All participants provided written informed consent, and the study was approved by the institutional ethics committee.
Patient evaluations
Baseline assessments included medical history, physical examination, N-terminal prohormone B-type natriuretic peptide (NT-proBNP) measurement, 6-minute walk distance (6MWD) test, computed tomography pulmonary angiography, and transthoracic echocardiography. Functional status and quality of life were assessed using the NYHA classification and the Kansas City Cardiomyopathy Questionnaire19.
Right heart catheterisation and pulmonary artery angiography
RHC was performed via the femoral vein immediately before PADN. Pulmonary artery angiography was performed using a 6 Fr pigtail catheter in the left anterior oblique 20°+cranial 20° or anteroposterior+cranial 20° projection (Figure 1A, Figure 1B). Imaging was performed without repositioning the angiographic table.
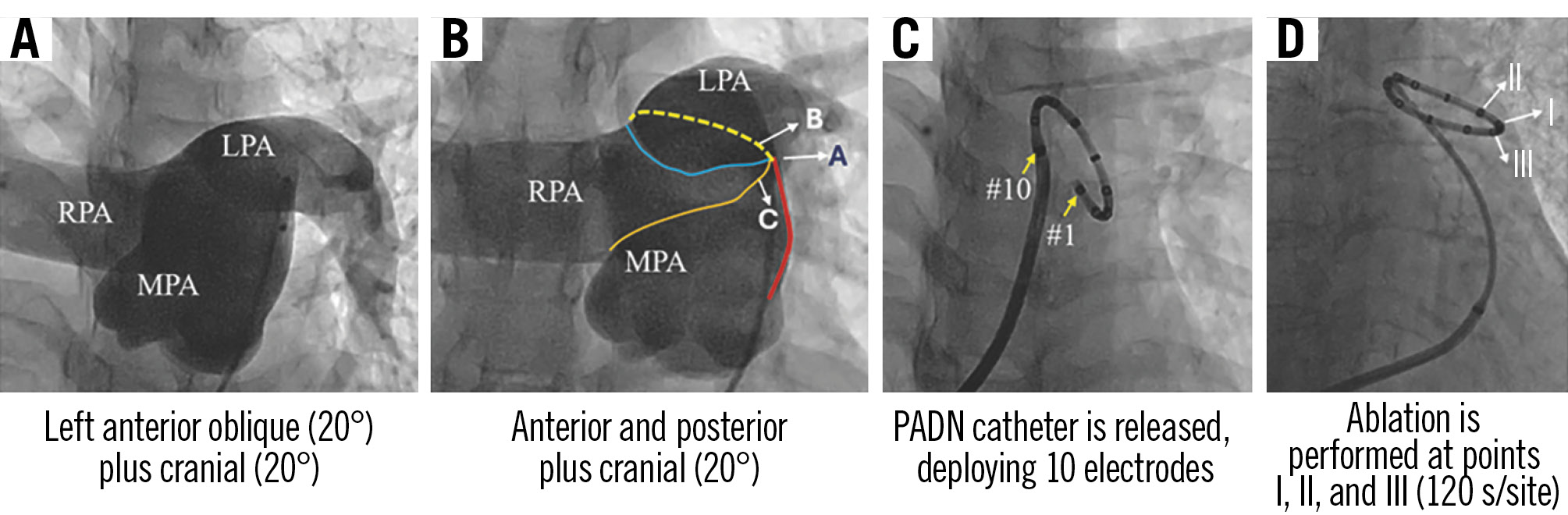
Figure 1. Pulmonary arterial angiography and PADN procedure. A) A clear view of the ostium of the LPA. B) The primary projection for PADN. C) The #1 electrode is seen distally, the #10 proximally. D) I, II, and III indicate the locations of panels A, B, and C, respectively. LPA; left pulmonary artery; MPA: main pulmonary artery; PADN: pulmonary artery denervation; RPA: right pulmonary artery
PADN device system
The PADN system consists of a radiofrequency generator, a control handle, and a PADN catheter (117 cm length), which includes the following (Figure 2): (1) a circular ablation segment with 10 preloaded electrodes (numbered 1-10), adjustable from 20-50 mm in diameter; (2) a shaft segment for curvature adjustment; (3) a red-coded diameter indicator for size selection; (4) a rotating control handle for precise positioning; and (5) a direct connection socket to the generator.
Each electrode is equipped with temperature, impedance, and energy monitoring wires (30 wires in total). A steering wire enables fine catheter control.
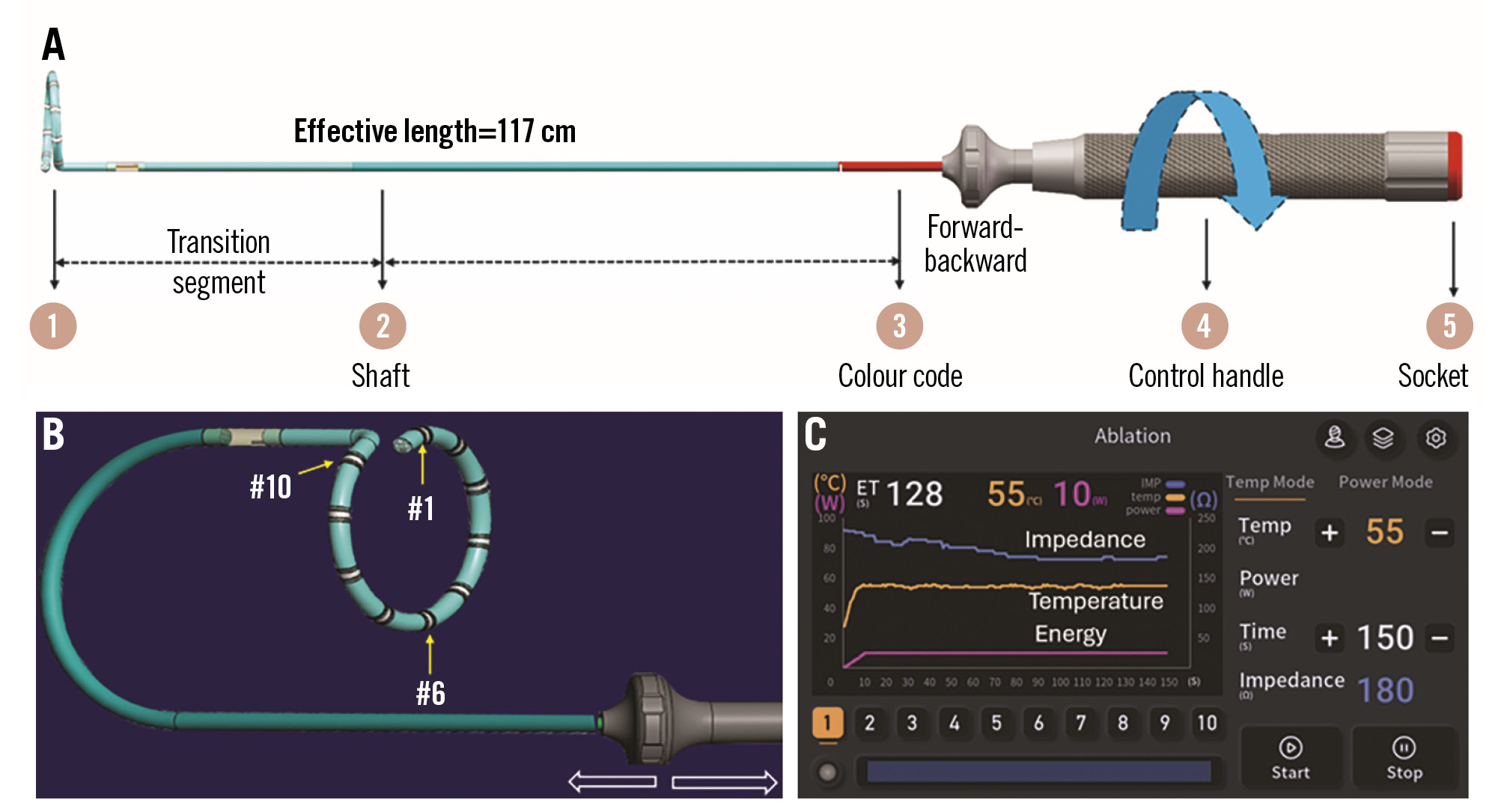
Figure 2. Description of PADN device. A) The PADN catheter; (B) electrode placement; (C) the display screen of the generator. PADN: pulmonary artery denervation
Defining ablation sites
Angiography in the anteroposterior-cranial projection (Figure 1B) identified anatomical landmarks: (1) red line: lateral wall of the main pulmonary artery; (2) blue line: anterior wall of the left pulmonary artery ostium; (3) yellow dashed line: posterior wall of the left pulmonary artery ostium; (4) orange line: anterior wall of the right pulmonary artery ostium. Three ablation sites were identified: point I, junction of all four lines; point II, 2 mm posterior to point I along the left pulmonary artery ostium; point III, 2 mm anterior to point I along the main pulmonary artery.
Following angiography, an 8.5 Fr long sheath was advanced into the left pulmonary artery. The PADN catheter was positioned via gentle pushing and rotation, aligning the electrodes with the defined ablation points (Figure 1C, Figure 1D). The ablation parameters were defined as a target temperature of ≥50°C, a maximum temperature of 60°C (automatic shutoff for safety), and an ablation duration per site of 120 seconds (maintaining 50±2°C).
Haemodynamic monitoring and dP/dt measurements
Haemodynamic waveforms were acquired at 2,000 samples/sec using LabChart and analysed via MATLAB, V2014 (MathWorks)19. Heart rate and beat-to-beat haemodynamics were processed using clustering techniques to exclude artefacts. Measurements included systolic/diastolic pressures from ventricular and aortic waveforms, mean arterial pressure from end-diastolic to subsequent end-diastolic intervals, and the maximum and minimum rates of change of ventricular pressure (dP/dtmax and dP/dtmin, respectively; filtered to remove valve closure artefacts). Haemodynamic data were obtained before PADN and monitored continuously for 10 minutes post-procedure.
Ventricular dP/dt was calculated using the sample-by-sample difference of the raw ventricular pressure waveform. For the maximum positive and maximum negative dP/dt measurements, the raw dP/dt waveform was smoothed first using a 9-sample median filter to remove valve closure artefacts, and then with a 30 Hz Butterworth low-pass filter before the maximum and minimum values were taken. After haemodynamic measurements were made for each beat, measurements taken from preventricular contractions, beats following preventricular contractions, as well as other artefacts such as measurements made during catheter repositioning, were eliminated using clustering techniques20.
Medications
Patients without atrial fibrillation received dual antiplatelet therapy (aspirin 100 mg/day and clopidogrel 75 mg/day) for 1 month. Those with pre-existing atrial fibrillation were maintained on oral anticoagulation.
Echocardiographic measurements and follow-up
Left ventricular (LV) volumes and LVEF were measured using Simpson’s biplane method. Tricuspid annular plane systolic excursion (TAPSE) was used to estimate right ventricular function, with a reference value for normal right ventricular systolic function set at 16 mm or more. TAPSE was obtained by placing the M-mode cursor through the right ventricular apex and the lateral aspect of the tricuspid annulus in the four-chamber view and measuring the amount of longitudinal motion of the annulus at peak systole. Other LV functional measurements were performed according to the previous report17.
Statistical analysis
Continuous variables are presented as mean±standard deviation. Paired t-tests or Wilcoxon signed-rank tests were used for within-group comparisons. Binary data were analysed using Fisher’s exact test. Haemodynamic changes were assessed at baseline and at 1, 3, 5, and 10 minutes post-PADN. A p-value<0.05 was considered statistically significant. Analyses were performed using SPSS, version 24.0 (IBM).
Results
Baseline characteristics
Between 26 March 2024 and 28 October 2024, ten patients (mean age: 65.4 years) with IpcPH secondary to HFpEF were enrolled (Table 1, Table 2). All patients were classified as NYHA Functional Class III at baseline. Two had ischaemic heart failure, and seven had chronic atrial fibrillation, of whom four underwent ablation; however, none maintained sinus rhythm.
Medications were administered per current guidelines, with seven patients (70%) receiving a combination of diuretics and SGLT2 inhibitors. At baseline, seven (70%) were on warfarin or direct oral anticoagulants. Two patients had undergone surgical valve replacement (one aortic, one mitral) 7 months prior to enrolment.
The mean 6MWD was 357±65 m, and NT-proBNP levels were 669±213 pg/mL; the mean LVEF was 60.8% (Table 2). Echocardiography revealed LV dilation (52.1±5.7 mm), increased left atrial volume index (45.8±3.6 mL/m²), and right atrial enlargement (54.1±7.7 mm). RHC confirmed that all patients met HFpEF criteria, with elevated mean PAP (24.2±2.3 mmHg) and PAWP (17.5±1.7 mmHg) but preserved PVR (<2 Wood units) (Table 2).
Table 1. Baseline clinical and echocardiographic characteristics.
| Results (N=10) | |
|---|---|
| Demographics | |
| Age, years | 65.4±9.5 |
| Male sex | 6 (60) |
| Body mass index, kg/m2 | 26.2±6.6 |
| Hypertension | 6 (60) |
| Diabetes mellitus | 5 (50) |
| Hyperlipidaemia | 3 (30) |
| Coronary artery disease | 2 (20) |
| Previous myocardial infarction | 1 (10) |
| Previous coronary intervention | 1 (10) |
| Atrial fibrillation | 7 (70) |
| Post-ablation | 4 (40) |
| Chronic | 5 (50) |
| Paroxysmal | 2 (20) |
| NYHA Functional Class III | 10 (100) |
| 6-minute walk distance, m | 357±65 |
| NT-proBNP, pg/mL | 669±213 |
| Medical and device therapy | |
| b-blockers | 1 (10) |
| ACE inhibitors or ARBs | 1 (80) |
| Furosemide | 8 (80) |
| Spironolactone | 10 (100) |
| SGLT2 inhibitors | 9 (90) |
| Combination of medications above* | 7 (70) |
| Warfarin | 1 (10) |
| New oral anticoagulants | 6 (60) |
| Defibrillator | 0 (0) |
| Cardiac resynchronisation therapy | 0 (0) |
| Surgical aortic valve replacement | 1 (10) |
| Transcatheter edge-to-edge repair | 0 (0) |
| Surgical replacement of mitral valve | 1 (10) |
| Echocardiographic measurements | |
| Left ventricular ejection fraction, % | 60.8±3.5 |
| LV end-diastolic diameter, mm | 52.1±5.7 |
| LV end-systolic diameter, mm | 34.8±3.7 |
| Left atrial diameter, mm | 47.4±3.9 |
| Left atrial volume index, mL/m2 | 45.8±3.6 |
| E/e ratio, % | 11.6±4.5 |
| Right atrial long-axis diameter, mm | 54.1±7.7 |
| Right atrial short-axis diameter, mm | 43.8±7.6 |
| Right ventricular TAPSE, mm | 16.8±0.7 |
| Right ventricular FAC, % | 36.6±2.1 |
| Data are n (%) or mean±SD. *Denotes the combination of diuretics and SGLT2 receptor inhibitors. ACE: angiotensin-converting enzyme; ARB: angiotensin II receptor blocker; eGFR: estimated glomerular filtration rate; FAC: fractional area change; LV: left ventricular; NT-proBNP: N-terminal prohormone B-type natriuretic peptide; NYHA: New York Heart Association; SD: standard deviation; SGLT2: sodium-glucose co-transporter-2; TAPSE: tricuspid annular plane systolic excursion | |
Table 2. Haemodynamic measurements via right heart catheterisation.
| Baseline (n=10) | At 10 minutes after PADN (n=10) | p-value | |
|---|---|---|---|
| Right heart catheterisation | |||
| Systolic pulmonary arterial pressure, mmHg | 38.9±4.6 | 32.3±6.4 | 0.005 |
| Diastolic pulmonary arterial pressure, mmHg | 16.9±2.7 | 13.3±4.1 | 0.016 |
| Mean pulmonary arterial pressure, mmHg | 24.2±2.3 | 20.1±3.9 | 0.003 |
| Pulmonary arterial wedge pressure, mmHg | 17.5±1.7 | 13.5±3.4 | 0.008 |
| Cardiac output, L/min | 5.4±1.1 | 5.4±1.5 | 1.000 |
| Cardiac output index, L/min/1.73 m2 | 2.8±0.2 | 3.4±0.5 | 0.153 |
| Right atrial pressure, mmHg | 8.4±2.1 | 8.3±2.1 | 0.766 |
| Pulmonary vascular resistance, Wood units | 1.3±0.4 | 1.4±0.6 | 0.801 |
| dP/dt ratio of left ventricle, mmHg/s | |||
| Maximum value | 1,685±500 | 1,778±43 | 0.262 |
| Minimum value | –1,699±323 | –2,048±442 | 0.012 |
| Left ventricular ESP, mmHg | 151±10 | 158±13 | 0.187 |
| Heart rate, beats/minute | 76±8 | 76±9 | 0.947 |
| Data are mean±SD. dP/dt: change of pressure over time; ESP: end-systolic pressure; PADN: pulmonary artery denervation; SD: standard deviation | |||
PADN procedure
The PADN procedure was successfully performed in all patients without procedural complications. The mean procedure time was 62 minutes (range: 45-75 minutes), including RHC, pulmonary artery angiography, PADN, and femoral/jugular venous haemostasis. All patients experienced mild chest discomfort during the procedure, but none required sedation. All were discharged the following day without complications.
Dynamic haemodynamic changes
At 10 minutes post-PADN, systolic PAP decreased from 38.9±4.6 mmHg to 32.3±6.4 mmHg (a 16.9% reduction) (Table 2, Figure 3A), with mean PAP and PAWP reductions of 16.9% and 22.9%, respectively. No significant changes were observed in cardiac output, right atrial pressure, or PVR.
A mean PAP reduction was observed in all ten patients (Figure 3B). However, one patient (a 54-year-old male with a history of surgical aortic valve replacement 20 years prior) exhibited a transient increase in PAWP at 10 minutes post-PADN (Figure 3C). This coincided with a 31.9% increase in cardiac output. Repeat RHC at 24 hours post-PADN showed a PAWP decrease to 12 mmHg, with cardiac output declining from 6.2 L/min to 5.0 L/min.
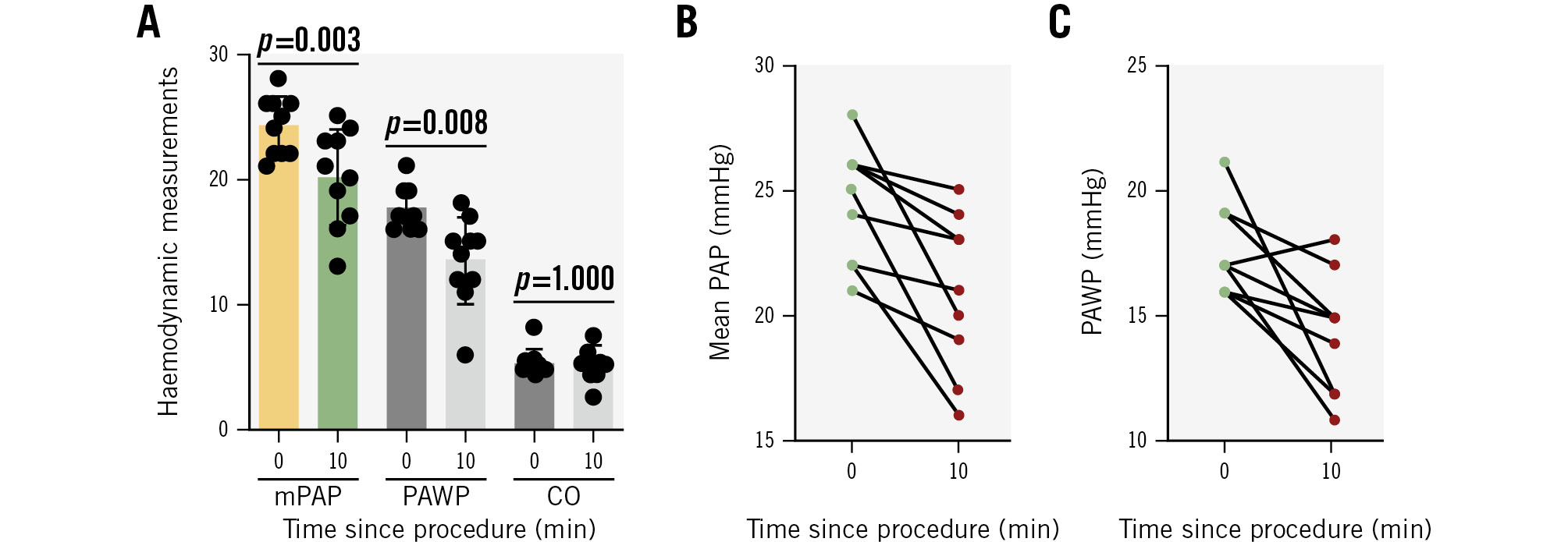
Figure 3. Dynamic haemodynamic changes. A) Haemodynamic changes over 10 minutes; (B) PAP changes in individuals; (C) PAWP changes in individuals. CO: cardiac output; mPAP: mean pulmonary arterial pressure; PAP: pulmonary arterial pressure; PAWP: pulmonary artery wedge pressure
Continuous monitoring of dP/dt
Monitoring of dP/dt was conducted in all patients (Figure 4). At baseline, dP/dtmax was 1,684.8±500.5 mmHg/s, with no significant change at 10 minutes post-PADN (1,777.5±433.2 mmHg/s; p=0.262) (Table 2). This was consistent with the absence of significant change in left ventricular end-systolic pressure (LVESP, baseline: 151.4±10.2 mmHg vs 157.7±12.5 mmHg post-PADN; p=0.187).
The minimum dP/dt significantly decreased from –1,698.9±322.9 mmHg/s at baseline to –2,048.0±442.3 mmHg/s at 10 minutes post-PADN (20.5% reduction; p=0.012) (Central illustration). This reduction became significant at 1 minute (10.7% reduction) after the PADN procedure.
Of the ten patients, two (both with prior aortic or mitral valve replacement) experienced an increase in dP/dtmin at 10 minutes post-PADN, alongside a significant rise in cardiac output (from 5.2 L/min to 7.3 L/min in one patient and from 4.4 L/min to 7.6 L/min in the other). By 24 hours post-procedure, cardiac output had returned to near-baseline levels (5.3 L/min and 4.6 L/min, respectively) without subsequent increases in the mean PAP or PAWP.
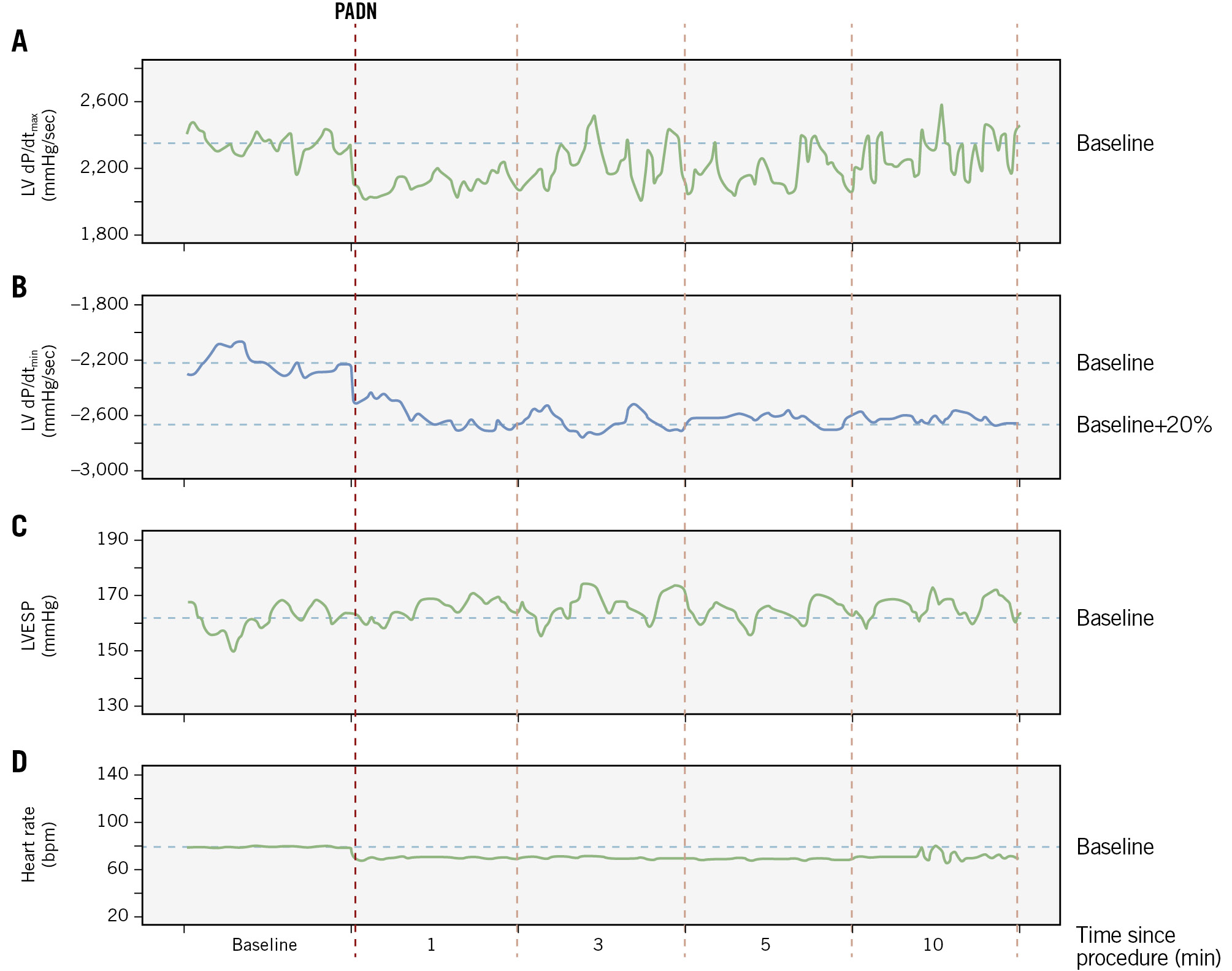
Figure 4. Dynamic changes of dP/dt, LVESP, and heart rate. A) No significant change in maximal LV dP/dt; (B) a significant reduction in minimal LV dP/dt; (C) no significant change in LVESP; (D) the heart rate remained unchanged over the 10-minute period. dP/dt: change of ventricular pressure over time; LV: left ventricular; LVESP: LV end-systolic pressure; PADN: pulmonary artery denervation
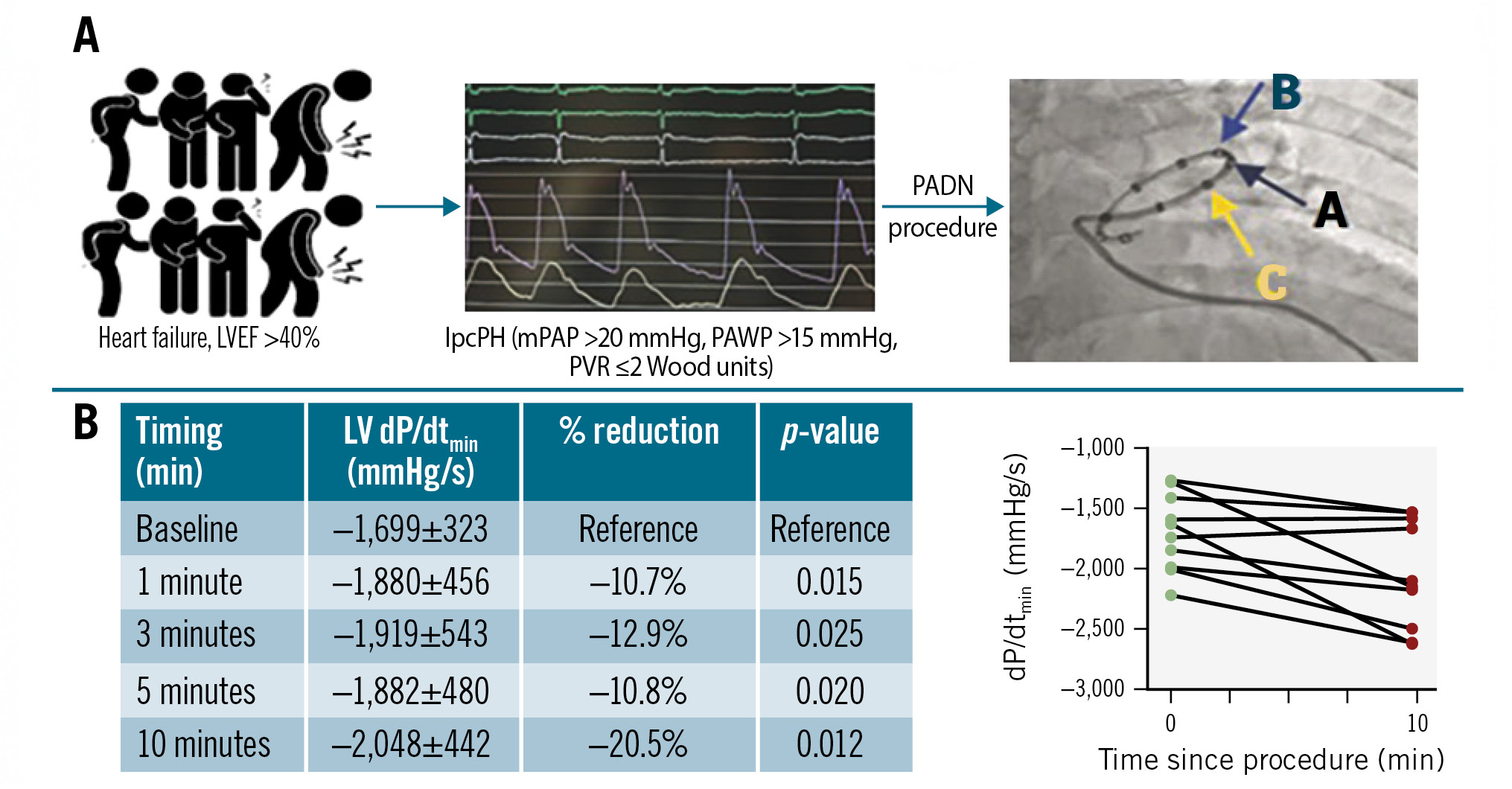
Central illustration. Dynamic change of dP/dt ratio during 10-minute monitoring. A) Study flowchart. B) Change in minimal dP/dt over time. dP/dt: change of ventricular pressure over time; IpcPH: isolated post-capillary pulmonary hypertension; LV: left ventricular; LVEF: left ventricular ejection fraction; mPAP: mean pulmonary arterial pressure; PADN: pulmonary artery denervation; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance
Discussion
This study demonstrates, for the first time, the feasibility, safety, and potential clinical efficacy of PADN in patients with HFpEF and IpcPH. The PADN procedure was successfully and safely performed in all patients, leading to improved haemodynamic and functional outcomes. Notably, LV diastolic relaxation significantly improved, as indicated by a profound reduction in dP/dtmin.
The diagnostic threshold for PH in HF has been revised, with the mean PAP cutoff reduced from 25 mmHg to 20 mmHg20. Current guidelines define IpcPH as a PVR ≤2 Wood units3. Since pulmonary vasodilators recommended for WHO Group I pulmonary arterial hypertension are not approved for PH-HF910, GDMT remains the standard of care for HFrEF but not for HFpEF. Consequently, device-based therapies have emerged as promising alternatives for this high-risk population. Over the past decade, transcatheter atrial shunt therapy, which creates an interatrial passage to unload left atrial pressure, has been investigated as a novel treatment for HF2122. However, optimal shunt sizing, HF phenotype selection, and long-term clinical outcomes remain unresolved challenges.
HF is characterised by autonomic dysfunction, marked by sympathetic overactivation and parasympathetic withdrawal. In the pulmonary circulation, excessive sympathetic activity contributes to increased PVR, PAP, and RV dysfunction12131723. The mechanisms of pulmonary sympathetic overactivation in HF are multifactorial2324, involving heightened sympathetic outflow, β- and α-adrenergic dysregulation, baroreceptor and chemoreceptor dysfunction, neurohormonal activation (renin-angiotensin-aldosteron system [RAAS] and arginine vasopressin pathways), and RV afterload and dysfunction. The PADN-5 study demonstrated that PADN improves exercise capacity, cardiac function, and clinical outcomes in patients with CpcPH, regardless of HF phenotype (HFrEF vs HFpEF)17. However, its role in HFpEF-induced IpcPH remains unexplored.
IpcPH is characterised by elevated pulmonary artery pressures due to passive backward transmission of left atrial pressure, without significant pulmonary vascular remodelling. Consequently, pulmonary vasodilators are not recommended, as they may exacerbate pulmonary congestion by increasing left-to-right shunting172224. In this study, PADN resulted in a significant reduction in PAP, with a concomitant 16.9% reduction in PAWP. Importantly, no patient developed pulmonary oedema, likely due to the simultaneous lowering of PAP and improved blood return post-procedure. This suggests PADN’s potential in addressing LV diastolic dysfunction in HFpEF-induced IpcPH.
Traditionally, neurohormonal crosstalk between the heart and the systemic circulation has been considered central to HF pathophysiology. However, neural mechanisms may be even more critical than circulating hormones in regulating cardiac function25. In this study, only one patient received a β-blocker, reinforcing the idea that the observed haemodynamic benefits were attributable to PADN rather than pharmacological therapy. Similarly to PADN-5 findings, our study revealed a profound reduction in PAWP (–22.9%), which correlates with improved LV relaxation. HFpEF is primarily driven by impaired LV diastolic compliance, even in the presence of preserved LVEF (>40%)421. In cardiac physiology, dP/dtmax is an indicator of LV contractility, while dP/dtmin reflects LV relaxation26. Notably, we observed (1) no significant change in dP/dtmax or LVESP over 10 minutes post-PADN and (2) a significant 20.5% reduction in dP/dtmin, mirroring the PAWP reduction and strongly suggesting improved LV relaxation.
Interestingly, two patients with prior surgical valve replacement exhibited transient (<24 hours) increases in dP/dtmin and cardiac output post-PADN, which subsequently normalised without adverse haemodynamic consequences. This suggests that cardiac output fluctuations may transiently affect dP/dtmin – an area warranting further investigation.
Limitations
This study has several limitations. The small sample size and non-normal data distribution limit generalisability, necessitating a larger, multicentre study. The short follow-up duration (10 minutes post-PADN) precludes assessment of long-term clinical effects. However, our ongoing nationwide, multicentre, randomised trial (ClinicalTrials.gov: NCT05824923) will provide 6-month follow-up data next year.
Finally, neurohormonal biomarkers (RAAS, noradrenaline) were not measured, as this study primarily aimed to evaluate PADN’s feasibility and its effect on LV relaxation.
Conclusions
This study provides initial evidence supporting the feasibility and safety of PADN in HFpEF-induced IpcPH. PADN was associated with significant improvements in LV relaxation and a subsequent reduction in PAWP, highlighting its potential as a novel device-based treatment for HFpEF with IpcPH.
Impact on daily practice
Heart failure (HF) remains a leading cause of mortality, with HF with preserved ejection fraction (HFpEF) accounting for a substantial proportion of cases. Isolated post-capillary pulmonary hypertension (IpcPH) in HFpEF represents an advanced disease state, with progressive pulmonary hypertension (PH) and impaired left ventricular (LV) relaxation. Current treatment options for HFpEF are limited to diuretics and sodium-glucose cotransporter-2 inhibitors, as no approved PH therapies have demonstrated efficacy in IpcPH. Pulmonary artery denervation (PADN) has demonstrated efficacy in combined post-capillary and precapillary PH but has not been studied in HFpEF-induced IpcPH until now. This feasibility study suggests that PADN leads to significant reductions in pulmonary arterial pressure and pulmonary artery wedge pressure (PAWP). PAWP reduction correlates with improved LV relaxation, as evidenced by significant reductions in the minimum rate of change of LV pressure. Further randomised trials are warranted to assess PADN’s long-term clinical benefits in HFpEF with IpcPH.
Acknowledgements
We appreciate all staff who participated in data collection and remote monitoring.
Funding
This trial was funded by the Jiangsu Provincial & Nanjing Municipal Clinical Trial Project (grant number BE 2019615).
Conflict of interest statement
S.-L. Chen reports speaker fees from MicroPort, Pulnovo, Boston Scientific, Medtronic, Sanofi, and BioMed; and grants from the National Scientific Foundation of China. The other authors have no conflicts of interest to declare. The Guest Editor reports receiving fees from Terumo.
