The introduction of drug-eluting stents (DES) has significantly altered the treatment of coronary artery disease. The critical differences in the new stent’s era were drug-coating advancements and decreased strut thickness1. Compared with bare metal stents (BMS), DES have ameliorated the outcomes of percutaneous coronary intervention (PCI), mainly by suppressing smooth muscle cell proliferation and subsequent neointimal hyperplasia, the main contributor of in-stent restenosis (ISR). Despite the advancements and innovations in the field, the incidence of ISR remains relatively high2. During the past decade, ISR-related interventions in the United States have consistently remained around 1/10 of all PCI, while the rate for target lesion revascularisation (TLR) hovers at 1-2% per year3. The treatment of ISR is a field of extensive research, with DES and drug-coated balloons (DCBs) being its main options. Considering the disadvantages of implanting an additional DES, DCBs have a notable convenience, since they can provide the remedy without the need for an additional stent to be implanted. However, until now, experts have recommended the use of DES as a first choice for treating ISR, leaving the use of DCBs to the discretion of the operator after considering several anatomical and procedural aspects45.
There is a significant interest in developments in DCB technology; specifically, trying to avoid a second stent layer and increased incidence of recurrent ISR. DCBs have proven highly effective in treating ISR, with the most widely available being paclitaxel-coated balloons (PCBs) and limus DCBs. Paclitaxel is a well-established antiproliferative agent known for its rapid cellular uptake due to its lipophilic nature45. Recent advancements in drug delivery technologies for limus DCBs have increased their use in clinical practice. New-generation sirolimus DCBs have shown promising results in recent trials, positioning them as a potential alternative to the widely used PCBs. Another emerging option is biolimus A9 (Biosensors International Group, Ltd.), a sirolimus derivative with enhanced lipophilicity compared to other limus agents, with retained rapamycin-inhibitory properties6. However, there is a lack of robust comparative data between limus DCBs versus PCBs on their efficacy and safety in treating ISR, making it difficult to generalise the results from individual studies.
The objective of this systematic review and meta-analysis was to compare the efficacy and safety of limus DCBs with the most extensively studied PCBs in patients with DES-ISR.
Methods
The present systematic review (SR) was performed according to the guidelines of the Cochrane Handbook for SRs7 and on the basis of a preregistered PROSPERO protocol (CRD42024583030). Reporting was performed based on the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) 2020 statement. A checklist of the reported PRISMA 2020 items is presented in Supplementary Table 1.
Information sources and search strategy
Systematic searches were conducted in MEDLINE (via PubMed), Scopus, and the Cochrane Library from inception up to 20 February 2025 without language restrictions. A search strategy was developed using the following search algorithm: [(Paclitaxel or Pacitaxel or Onxol or Taxane or Taxol or PTX or Abraxane) AND (Sirolimus or Rapamycin or mTOR or Rapamune or Zotarolimus or ABT-578 or Everolimus or Biolimus or Afinitor or Certican or RAD001) AND (balloon)]. Reference lists from selected studies and pertinent reviews and abstracts of international conferences from the last ten years were also scrutinised.
Eligibility criteria
Population and interventions
Adults (>18 years old) with clinical evidence of stable or unstable angina or a positive functional study and DES-ISR treated with a DCB were considered for enrolment. Eligible studies were required to compare any limus-based coated balloon – sirolimus- or biolimus-coated – with paclitaxel-coated balloons in terms of effectiveness and/or safety.
Outcomes
The primary outcomes were the incidence of clinically driven TLR and target lesion failure (TLF; defined as a composite of cardiac death, target vessel myocardial infarction [TVMI], and repeated – either surgical or percutaneous – TLR or clinically indicated TLR) during a one-year follow-up. Angiographic outcomes included in-segment and in-lesion late lumen loss (LLL; difference between the postprocedural and follow-up minimal lumen diameter [MLD], evaluated by quantitative coronary angiography), MLD, diameter stenosis (DS), and acute gain during the follow-up. Additional endpoints included procedural success (defined as the ability of balloon inflation to reach a <30% final stenosis, Thrombolysis in Myocardial Infarction 3 flow, no flow-limiting dissection, and freedom from in-hospital major adverse cardiovascular events [MACE]), MACE (occurrence of cardiac deathâ target vessel myocardial infarctionâ or clinically driven TLR) during follow-up, as well as individual clinical endpoints at 12-month follow-up (stent thrombosis, cardiac death, or target vessel myocardial infarction). Myocardial infarction (MI) was defined according to the established 4th Universal Definition of Myocardial Infarction8. Stent thrombosis, cardiac death, TVMI, and clinically driven TLR were defined according to the Academic Research Consortium consensus document9. Periprocedural complications were also recorded (dissections, perforations, slow-flow or no-reflow phenomena).
Study design
Eligible studies were randomised controlled trials (RCTs) of any duration including a minimum sample size of ten patients.
Exclusion criteria
Records were excluded based on the following criteria: (1) case reports/case series, narrative reviews; (2) expert opinions, dissertations, protocols; (3) studies including animals and/or in vitro studies.
Selection process
All records retrieved from the search strategy were screened by title and abstract by two investigators (M. Sagris and N. Ktenopoulos) independently. Subsequently, the same authors also independently screened the remaining studies’ full texts. Discrepancies at any stage were resolved by discussion or consultation with a third reviewer (S. Soulaidopoulos). The EndNote X7 software (Clarivate) was used for reference management.
Data extraction
A data extraction form was created, piloted, and standardised after discussion and calibration exercises. The following variables were extracted: first author; publication year; title; journal’s title; study type; distribution of randomised participants in each group; inclusion/exclusion criteria; demographic and population characteristics; comorbidities; interventions; comparator; angiographic, efficacy and safety outcomes; number of events; and number of patients in each group (priority to intention-to-treat [ITT] analysis).
Quality assessments
The methodological quality of the included studies was evaluated using the revised Cochrane risk of bias 2 (RoB 2) tool10 for RCTs. Methodological quality was evaluated by two authors independently, and any disagreements were resolved through discussion or involvement of a third author. Regarding the quality and risk of bias assessment, the criteria for the ascertainment of points and the allocation of points for each study are given in Supplementary Table 2.
Data analysis
All analyses were performed using Stata 16.0 (StataCorp). Frequencies with percentages (%) are presented for categorical variables, means with standard deviation for Gaussian continuous variables, and medians with interquartile range for non-normally distributed continuous variables. Summary estimates for categorical variables are reported as risk ratios (RRs), and for continuous variables, they are reported as the standardised mean difference (SMD). The RRs, SMDs and 95% confidence intervals (CIs) were calculated using the DerSimonian-Laird estimator for random-effects models. In case of zero events in a treatment arm, a continuity correction was applied. Heterogeneity was assessed using the I2 test and was formally tested with Cochran’s Q test. Rough cutoff values were 25%, 50%, and 75%, indicating low, moderate, and high heterogeneity, respectively11. Small-study effects (including publication bias) for each outcome were visually assessed with funnel plots and formally tested with the Egger’s test. For all statistical tests, a two-tailed p-value less than 0.05 was considered statistically significant.
Results
Study selection
A comprehensive literature search resulted in 1,245 potentially relevant records (864 in MEDLINE/PubMed, 198 in Cochrane Library and 183 in Scopus). Once duplicates were removed, 652 studies were identified. After screening titles and abstracts, 68 articles were retrieved for full-text evaluation. A total of six RCTs (five full articles1213141516 and one presented as a late-breaking clinical trial17) met the predetermined eligibility criteria and were included in this meta-analysis, as shown in the PRISMA flow diagram (Supplementary Figure 1). All studies were published in the last three years, from 2022 to 2024.
Baseline characteristics
A total of 639 versus 569 patients with ISR were included in the analysis in the limus DCB and PCB groups, respectively, with a mean follow-up of 12 months. The total number of treated lesions included 674 limus DCBs and 598 PCBs. The mean age of the limus DCB and PCB groups, respectively, was 66±9.8 years versus 65.8±9.9 years, and the majority of patients were male. Of the treated lesions, 3% (N=10/335)/1.5% (N=5/327) were located at the left main artery, 42.9% (N=209/487)/45.6% (N=219/480) at the left anterior descending artery, 20.3% (N=99/487)/17.3% (N=83/480) at the circumflex artery, and 33.2% (N=162/487)/35.2% (N=169/480) at the right coronary artery, in the limus DCB and PCB groups, respectively. Preprocedural examinations revealed a mean lesion length of 17.2±7.9 mm for limus DCBs versus 16±7.3 mm for PCBs. The mean maximum inflation pressure was 11±3.3 bar versus 11±4.4 bar, with a mean inflation duration of 58.4±7.6 seconds versus 58.4±9.4 seconds, for limus DCB and PCB groups, respectively. Full patient characteristics are presented in Table 1 and Table 2.
Table 1. Characteristics of the included studies.
| Study | Design | Arm A (limus DCB), N | Arm B (PCB), N | Lesions at baseline, N | Lesions at follow-up, N | Clinical/ angiographic follow-up, months | Lesion length, mm | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| B. Scheller 2022 (Malaysia)14 | RCT (SCB vs PCB) | 25 | 25 | 26 | 25 | 26 | 25 | 12 | 6 | 14.2±7.8 | 13.2±7.1 |
| B. Scheller 2022 (Europe)14 | RCT (SCB vs PCB) | 25 | 26 | 26 | 26 | 26 | 27 | 12 | 6 | 13.2±6.5 | 14.3±9.2 |
| C. Briguori 202313 | RCT (SCB vs PCB) | 186 | 186 | 186 | 186 | n/a | n/a | 12 | n/a | 19±5 | 18±6 |
| S. Fitzgerald 202317 | RCT (Biolimus DCB vs PCB) | 135 | 67 | 135 | 67 | 121 | 55 | 12 | 6 | 20.5±10.3 | 18.5±9.5 |
| H. Liu 20251633 | RCT (SCB vs PCB) | 130 | 128 | 149 | 141 | 149 | 141 | 12 | 9 | 13.4±7.5 | 12.7±6.4 |
| Y. Chen 202415 | RCT (Biolimus DCB vs PCB) | 138 | 137 | 152 | 153 | 114 | 116 | 12 | 9 | 16.2±7.2 | 15.9±6.8 |
| Values are N or mean±standard deviation. DCB: drug-coated balloon; N: number of patients; n/a: not available; PCB: paclitaxel-coated balloon; RCT: randomised controlled trial; SCB: sirolimus-coated balloon | |||||||||||
Table 2. Patient characteristics of the included studies.
| Study | Arm A (limus DCB), N | Arm B (PCB), N | Males, N | DM, N | HTN, N | DLP, N | LVEF, % | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B. Scheller 2022 (Malaysia)14 | 25 (SeQuenta) | 25 (SeQuent Pleasea) | 22 | 19 | 18 | 19 | 24 | 23 | 23 | 21 | 57±7 | 56±14 |
| B. Scheller 2022 (Europe)14 | 25 (SeQuenta) | 26 (SeQuent Pleasea) | 21 | 20 | 9 | 11 | 25 | 25 | 21 | 22 | 49±10 | 56±11 |
| C. Briguori 202313 | 186 (Devoirb) | 186 (RESTOREc) | 139 | 143 | 104 | 103 | 167 | 171 | 160 | 167 | 51±10 | 50±7 |
| S. Fitzgerald 202317 | 135 (Biosensors biolimus) | 67 (SeQuent Pleasea) | 110 | 51 | 42 | 17 | 117 | 53 | 112 | 55 | n/a | n/a |
| H. Liu 20251633 | 130 (SeQuenta) | 128 (SeQuent Pleasea) | 95 | 97 | 44 | 53 | 87 | 90 | 23 | 18 | 59.2±8 | 60.6±7.5 |
| Y. Chen 202415 | 138 (Biosensors biolimus) | 137 (SeQuent Please Neoa) | 103 | 101 | 51 | 59 | 86 | 98 | 61 | 53 | 61±8 | 60.8±9 |
| Values are N or mean±standard deviation. aBy B. Braun; bby APR Medtech; cby Cardionovum GmbH. DCB: drug-coated balloon; DLP: dyslipidaemia; DM: diabetes mellitus; HTN: hypertension; LVEF: left ventricular ejection fraction; N: number of patients; n/a: not applicable; PCB: paclitaxel-coated balloon | ||||||||||||
TLR and TLF outcomes
All six RCTs reported on TLR incidence, showing no statistical difference between the two groups of limus DCBs versus PCBs (14% vs 11.4%; RR 1.16, 95% CI: 0.82-1.64; I2=14%) at one-year follow-up. Four studies reported on TLF rates, also without statistical difference between the two groups (15% vs 14%; RR 1.14, 95% CI: 0.86-1.53; I2=0%). Low heterogeneity was observed among the studies (Figure 1).
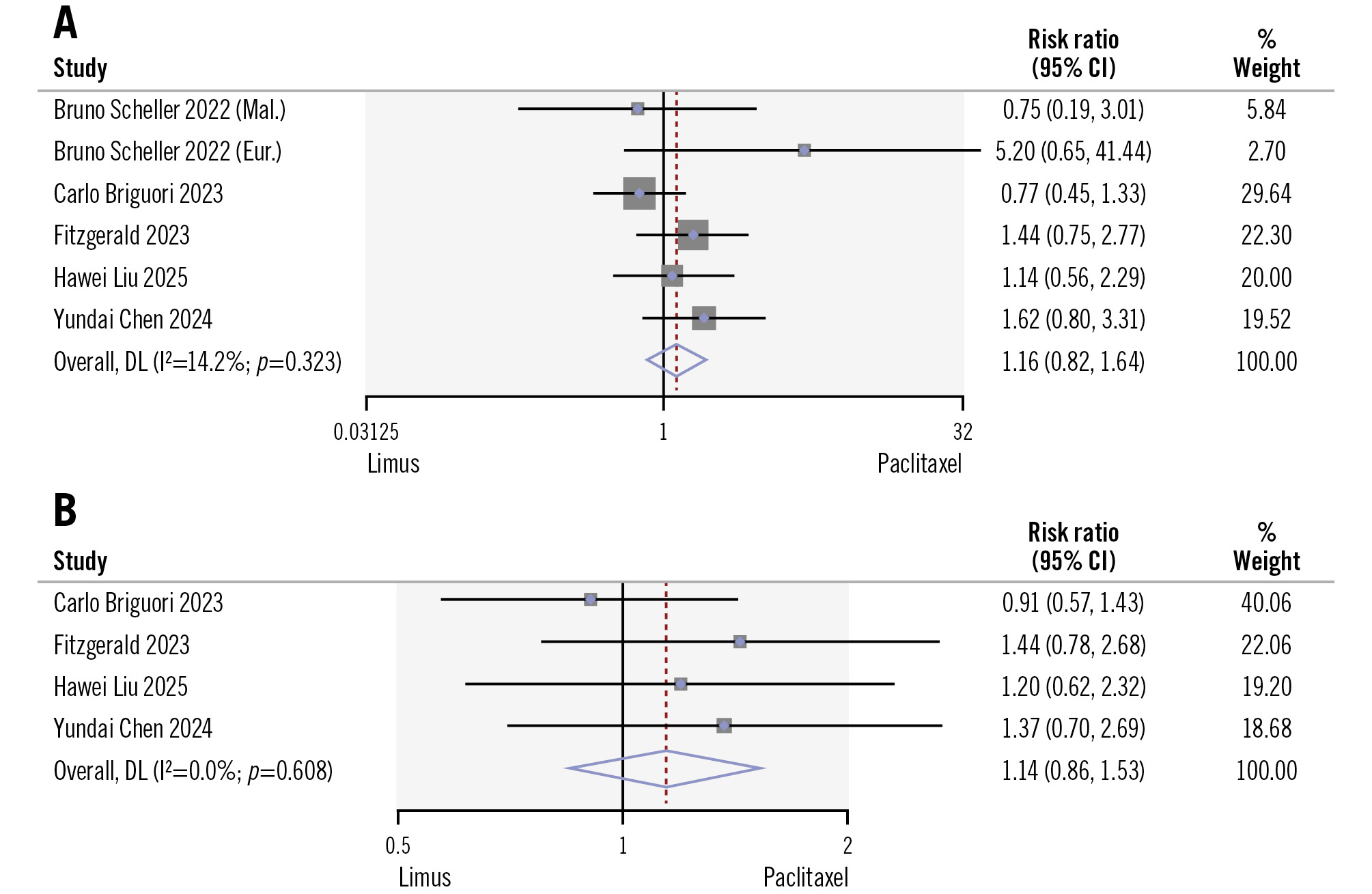
Figure 1. Comparison of limus DCBs versus PCBs in terms of TLR and TLF. A) TLR; (B) TLF. Study references are provided in Table 1 and Table 2. CI: confidence interval; DL: DerSimonian-Laird; DCB: drug-coated balloon; PCB: paclitaxel-coated balloon; TLF: target lesion failure; TLR: target lesion revascularisation
Procedural and clinical outcomes
Overall procedural success was similar between the two groups, with favourable outcomes in nearly all cases (limus DCB: 188/188 cases vs PCB: 187/188 cases). During the periprocedural/30-day post-intervention period, there was one case of dissection in the limus DCB group. There were no differences in the incidence of MACE (16.4% vs 13.5%; RR 1.11, 95% CI: 0.84-1.46; I2=0%) at one-year follow-up (Figure 2). More specifically, the pooled all-cause mortality (1.8% vs 1.4%; RR 1.25, 95% CI: 0.50-3.08; I2=0%) and cardiac death (1.4% vs 1%; RR 1.19, 95% CI: 0.42-3.41; I2=0%) rates during the one-year follow-up across the six studies showed no statistical differences between the limus DCB and PCB groups, respectively (Figure 2). Additionally, no differences were observed in TVMI (0.9% vs 1%; RR 0.58, 95% CI: 0.20-1.67; I2=0%) or any MI (1.8% vs 0.9%; RR 0.58, 95% CI: 0.20-1.67; I2=0%) (Figure 3). Finally, there were no statistical differences in the incidence of stent thrombosis (0% vs 0.3%; RR 0.44, 95% CI: 0.06-3.06; I2=0%) or the rates of any vessel revascularisation (8.7% vs 7.2%; RR 1.34, 95% CI: 0.91-1.98; I2=10%) during this period. Low heterogeneity was observed among the studies.
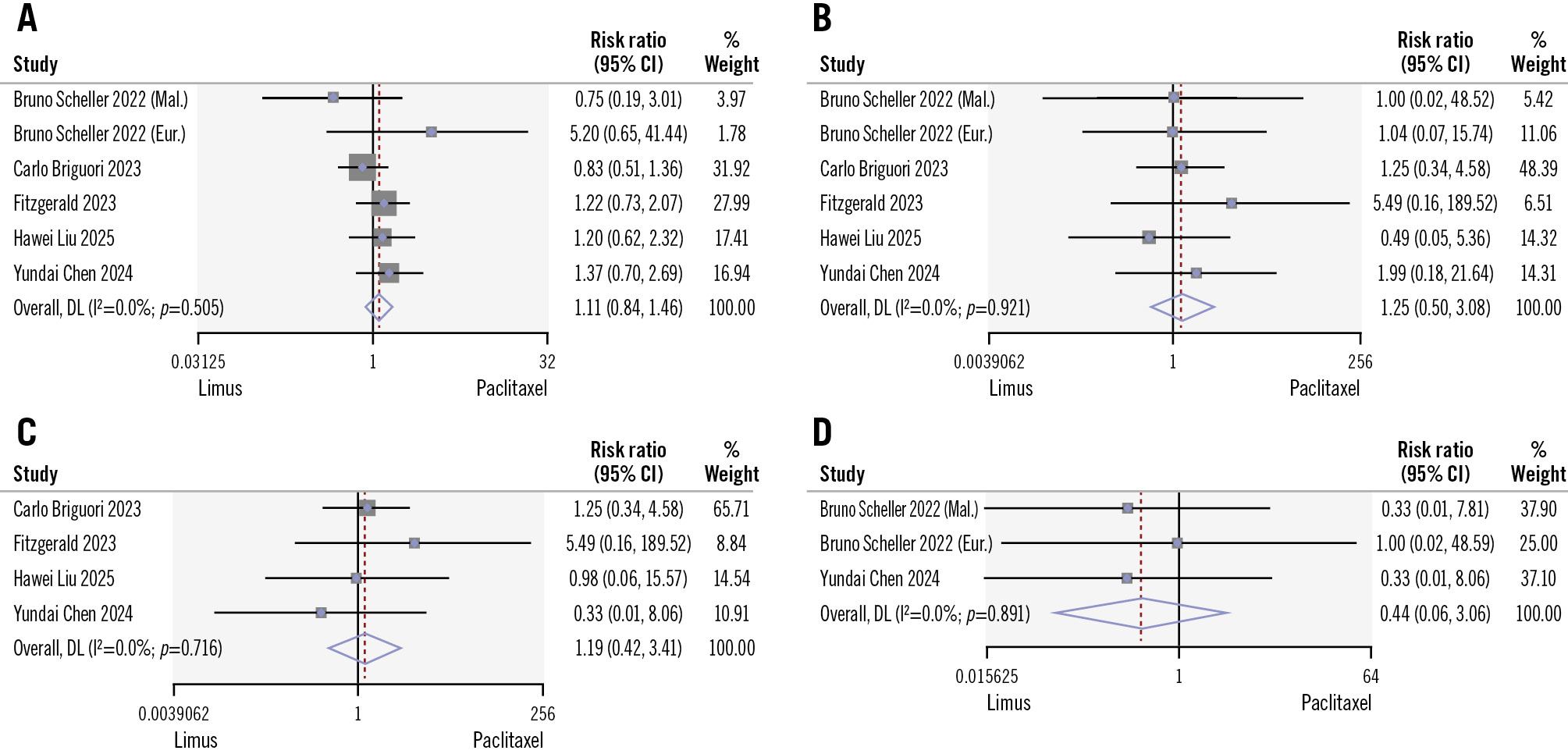
Figure 2. Comparison of limus DCBs versus PCBs in terms of MACE, all-cause mortality, cardiac death, and stent thrombosis. A) MACE; (B) all-cause mortality; (C) cardiac death; (D) stent thrombosis. Study references are provided in Table 1 and Table 2. CI: confidence interval; DL: DerSimonian-Laird; DCB: drug-coated balloon; MACE: major adverse cardiovascular events; PCB: paclitaxel-coated balloon
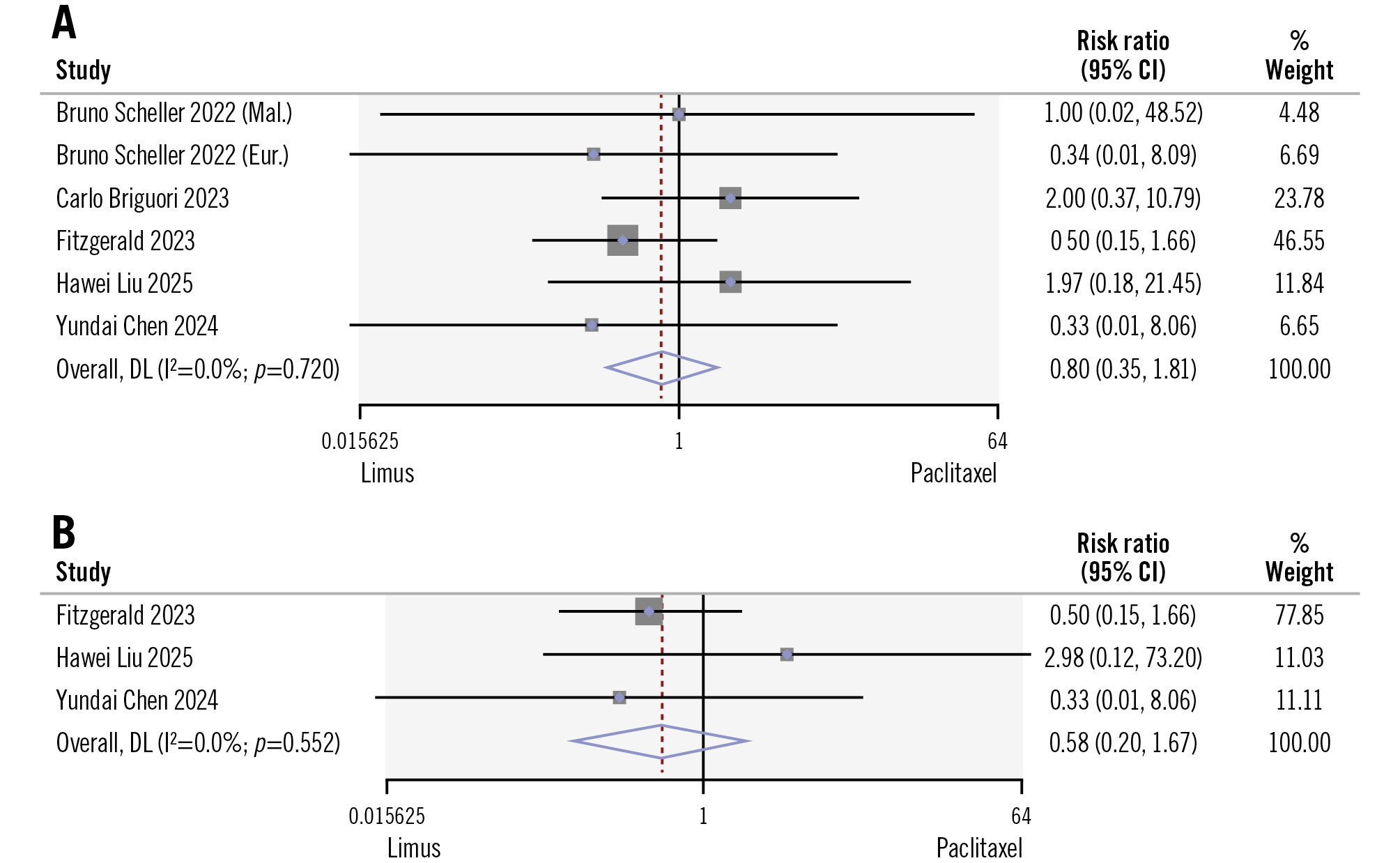
Figure 3. Comparison of limus DCBs versus PCBs in terms of myocardial infarction and target vessel myocardial infarction. A) Myocardial infarction; (B) target vessel myocardial infarction. Study references are provided in Table 1 and Table 2. CI: confidence interval; DL: DerSimonian-Laird; DCB: drug-coated balloon; PCB: paclitaxel-coated balloon
Angiographic outcomes
The mean angiographic follow-up for both arms was 7.2±1.6 months. There was no significant difference in binary restenosis (19.5% vs 12.9%; RR 1.45, 95% CI: 0.77-2.71; I2=63%) between the two groups during the follow-up (Figure 4). Based on the quantitative coronary angiography measurements, the mean minimal lumen diameter at baseline and follow-up were analysed, presenting an SMD of +3.30 (95% CI: 2.64-3.95; I2=90%) for the limus DCB group and +3.27 (95% CI: 2.69-3.85; I2=87%) for the PCB group. When comparing the mean MLD of the two groups at follow-up, data from five studies showed no significant difference between the limus DCB and PCB groups, with an SMD of +0.06 (95% CI: –0.07 to 0.18; I2=0%) (Figure 4). The mean DS was also analysed at baseline and follow-up, presenting an SMD of –3.77 (95% CI: –4.75 to –2.79; I2=96%) for the limus DCB group and –4.61 (95% CI: –5.15 to –4.08; I2=79%) for the PCB group. The acute lumen gain from four studies presented an SMD of +0.09 (95% CI: –0.06 to 0.23; I2=0%) (Supplementary Figure 2). The overall estimated mean DS from six studies (Supplementary Figure 3) at follow-up showed a favourable trend towards the PCB group, but this did not reach statistical significance, with an SMD of +0.21 (95% CI: –0.02 to 0.45; I2=71%).
Finally, the risk of LLL at follow-up (in-segment LLL: 0.4±0.54 mm vs 0.28±0.43 mm; in-lesion LLL: 0.27±0.48 mm vs 0.29±0.43 mm) was similar between the limus DCB and PCB groups, with an SMD of +0.02 (95% CI: –0.18 to 0.23; I²=70.6%) for in-segment LLL analysis and –0.03 (95% CI: –0.31 to 0.24; I²=0.0%) for in-lesion LLL analysis (Figure 5). An intragroup analysis was performed for the reference vessel diameter (RVD) at baseline and follow-up, with the limus DCB group presenting an SMD of +0.55 (95% CI: 0.28-0.82; I2=27%) and the PCB group an SMD of +0.31 (95% CI: –0.09 to 0.70; I2=62%). Comparing the two groups in terms of the follow-up RVD measurements, there was no significant statistical difference, with an SMD of +0.39 (95% CI: –0.22 to 1.01; I2=83%) (Supplementary Figure 4). Low heterogeneity was observed among the analysed studies.
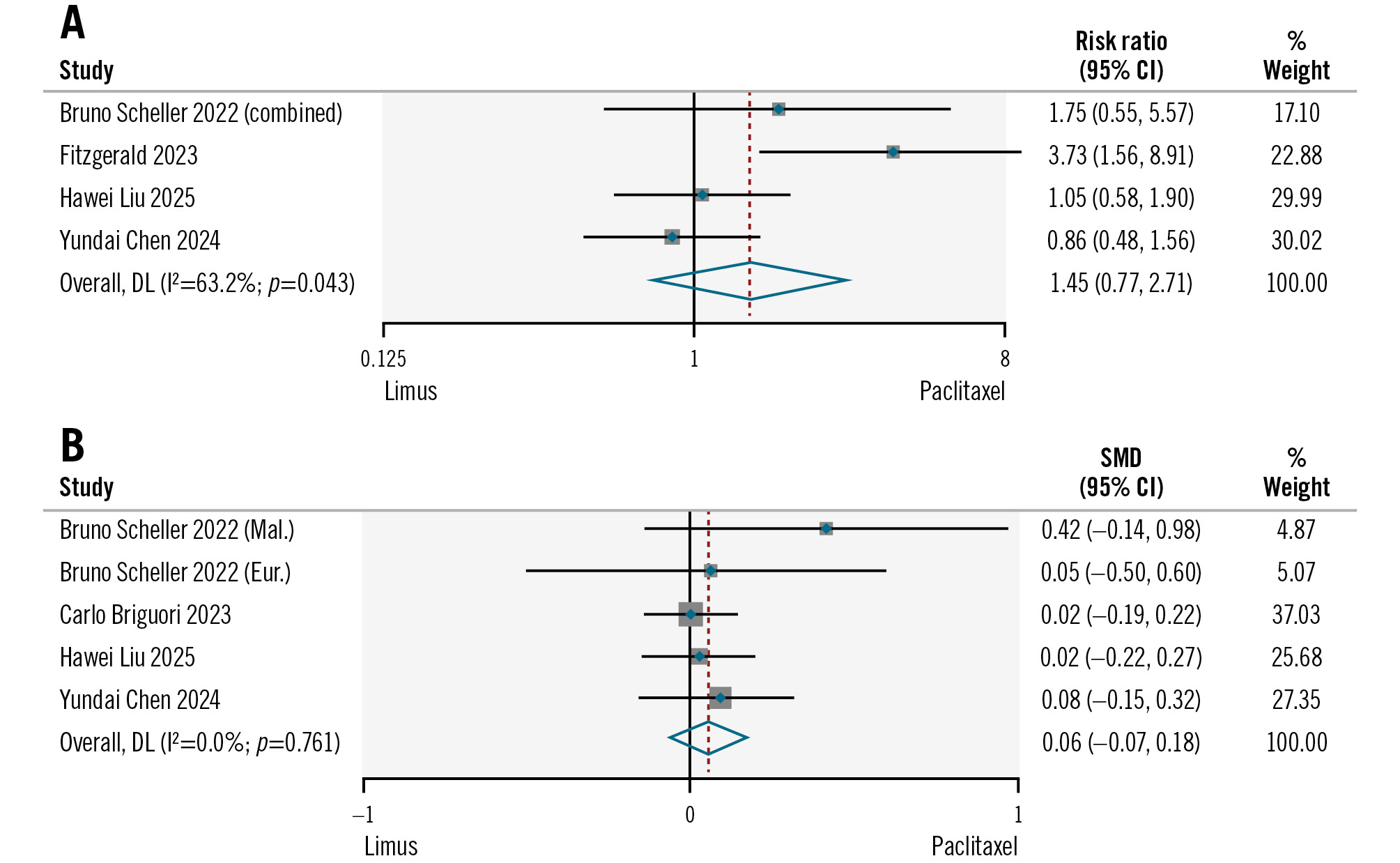
Figure 4. Comparison of limus DCBs versus PCBs in terms of binary restenosis during the follow-up and minimal lumen diameter post-intervention. A) Binary restenosis; (B) post-intervention minimal lumen diameter. Study references are provided in Table 1 and Table 2. CI: confidence interval; DL: DerSimonian-Laird; DCB: drug-coated balloon; PCB: paclitaxel-coated balloon; SMD: standardised mean difference
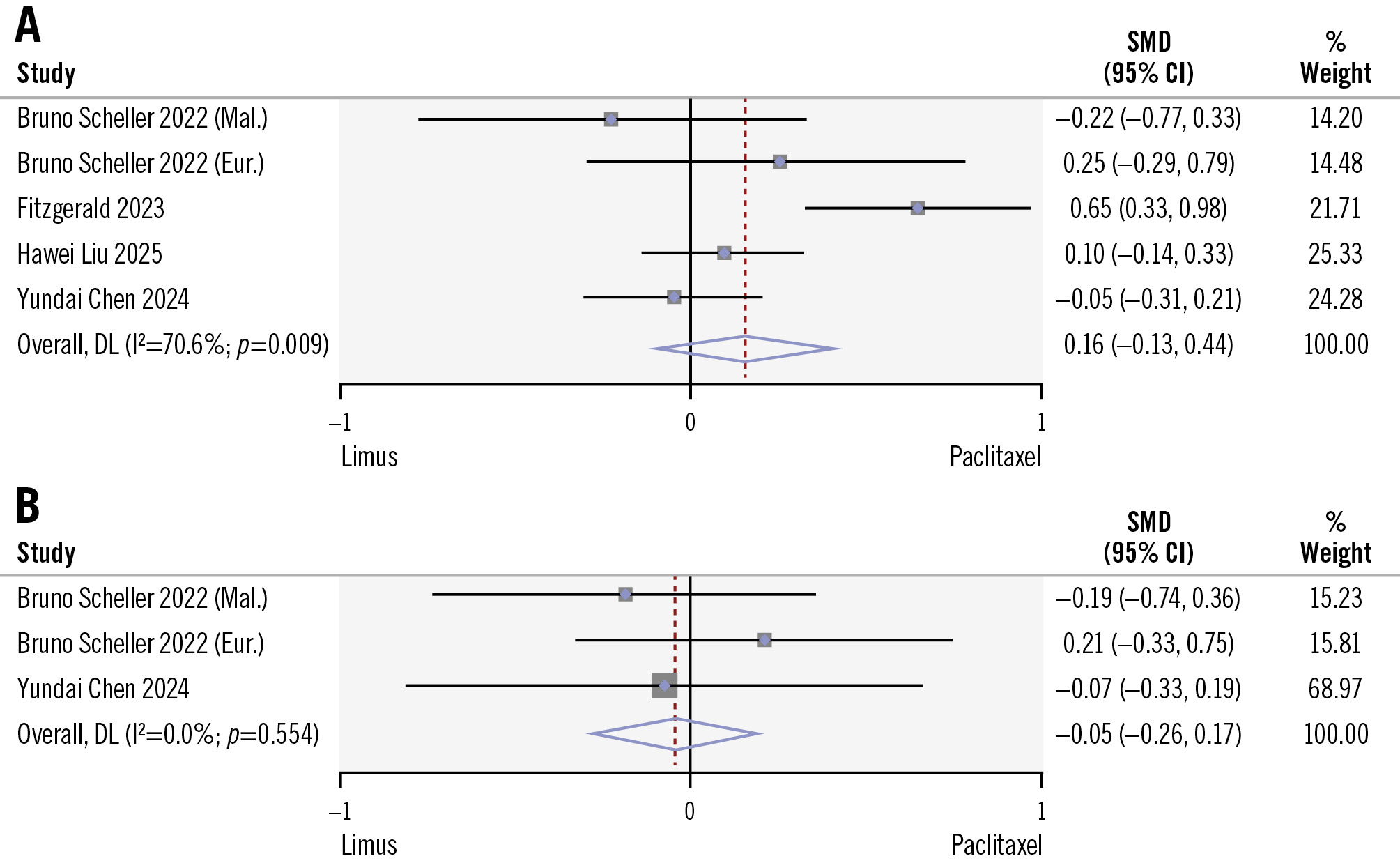
Figure 5. Comparison of limus DCBs versus PCBs in terms of in-segment and in-lesion late lumen loss during the follow-up. A) In-segment LLL; (B) in-lesion LLL. Study references are provided in Table 1 and Table 2. CI: confidence interval; DL: DerSimonian-Laird; DCB: drug-coated balloon; LLL: late lumen loss; PCB: paclitaxel-coated balloon; SMD: standardised mean difference
Discussion
This is a meta-analysis of 12-month follow-up data from six RCTs including a total of 1,208 patients comparing the main use of paclitaxel- to limus-coated balloons for the treatment of coronary ISR. Our findings indicate that there were no significant differences between PCBs and limus DCBs in the risk of major clinical outcomes, including all-cause mortality, cardiac death, and clinically driven TLR. Limus DCBs achieve at least equal angiographic outcomes compared to PCB without significant differences in LLL during follow-up (Central illustration).
In fact, the management of ISR in the DES era constitutes a considerable clinical and technical challenge, often necessitating revascularisation interventions5. Despite ongoing improvement in DES technologies, a substantial rate of ISR with current stent platforms ranging from 1-8% per year has been recorded18. To that end, several materials and techniques have been proposed for the optimal management of these lesions, including angioplasty with cutting or scoring balloons, DCBs, or placement of a new DES. The main advantage of DCBs is their ability to release an antiproliferative drug without leaving an additional metallic layer on the treated lesion. Although this concept seems quite attractive when treating ISR, where additional stent layers could increase the risk of new restenosis, the DAEDALUS analysis of 10 randomised trials demonstrated that DCB angioplasty and repeat stenting with DES are similarly effective and safe in the treatment of BMS-ISR, whereas DCB angioplasty was found to be less effective compared with DES implantation in the treatment of DES-ISR19. Nevertheless, DCBs could be the preferred option for less complex restenotic lesions, multilayer ISR cases (to avoid an additional metallic layer), BMS restenosis, and in patients at high bleeding risk to minimise the required duration of dual antiplatelet therapy5.
Until recently, paclitaxel was the drug of choice for most available DCBs. Owing to its lipophilic properties, paclitaxel rapidly penetrates the cell membrane, binds to the microtubules and inhibits their depolymerisation, thus irreversibly blocking cell division20. This mechanism explains the drug’s anti-inflammatory and antiproliferative effects. Limus-based drugs, on the other hand, although demonstrating higher safety and efficacy, are characterised by inferior lipophilicity compared with paclitaxel, a characteristic that poses a significant limitation to adequate drug distribution and retention when used in DCBs. However, technological advances have successfully overcome these challenges, with the encapsulation of the drug in a lipophilic nanocarrier leading to the development of limus DCBs with remarkable efficacy in the treatment of both de novo coronary lesions and ISR2021. As a result, initial doubts have gradually yielded to restrained enthusiasm regarding the use of limus-coated balloons for ISR, with a history similar to that of limus and paclitaxel DES22. In this meta-analysis, sirolimus- and biolimus-coated balloons were pooled into a single limus-coated balloon group. This approach is justified by their shared mechanism of action as mammalian target of rapamycin inhibitors, cytostatic effects on smooth muscle cell proliferation, and favourable endothelial healing profile compared with paclitaxel. The differences between sirolimus and biolimus are mainly in their pharmacokinetic properties (lipophilicity and tissue uptake) rather than their mechanisms.
Particularly for de novo coronary lesions, a recent meta-analysis demonstrated similar rates of clinically driven TLR and MACE with limus and paclitaxel DCBs among patients undergoing DCB-only PCI23. On the other hand, the risk of TLR seems to be higher in DCB-only PCI for ISR. The analysis of the one-year clinical follow-up of 642 patients enrolled in the EASTBOURNE registry, which evaluated the performance of a sirolimus DCB, showed a considerably higher incidence of TLR in patients treated for ISR compared with those with de novo coronary lesions (5.4 vs 0.2%; p=0.0008)24. Similar findings were reached in the retrospective analysis of 288 patients treated with the MagicTouch sirolimus-coated balloon (Concept Medical), with higher rates of TLR being observed among patients with ISR (17%) compared to those with de novo lesions (9%)25.
Multiple factors contribute to the higher rates of stenosis recurrence when treating ISR compared to de novo lesions. These are mainly procedural issues associated with the placement and condition of the existing stent. Studies using optical coherence tomography have identified several dominant predictors of adverse long-term outcomes when restenting ISR lesions. These include underexpansion of the older stent, the total amount of neoatherosclerotic or peristent calcium as well as the presence of multiple layers of stent struts26. Understanding the underlying mechanism of ISR by utilising intracoronary imaging techniques is of crucial importance for planning the optimal treatment strategy and choosing the proper devices for PCI. For instance, it is important to optimise the old stent’s expansion and apposition using balloon dilatation when detecting stent underexpansion and underdeployment as the possible cause of restenosis before proceeding to further treatment with a new stent or a DCB. Detection of severe neointimal hyperplasia or neoatherosclerosis as the primary cause of ISR may necessitate the use of debulking strategies prior to DCB or DES implantation, such as cutting and scoring balloons, which create small fissures in the atherosclerotic plaque, allowing for deeper drug penetration2728. Furthermore, advanced technologies such as intravascular lithotripsy and rotational atherectomy provide the potential for more aggressive plaque modification and have demonstrated promising results in the management of failed, underexpanded stents implanted in heavily calcified lesions or severely calcified in-stent neoatherosclerosis293031. All these interventions are essential for optimal lesion preparation before applying DCBs and stent implantation, ultimately contributing to improved long-term outcomes. Indeed, performance of optimised predilatation of the restenotic lesion appears to be a more significant predictor of TLR rates than the specific type of DCB used when treating ISR13.
To that end, factors related to optimised lesion preparation must be carefully considered when evaluating long-term angiographic outcomes in the comparison between PCBs and limus DCBs for the treatment of restenotic lesions. It is true that the methods utilised for successful lesion preparation are not thoroughly described in the included studies. Of note, though, neither of the two types of DCBs compared in this analysis was associated with worse angiographic outcomes. This comes in contrast with a recent pooled analysis of trials comparing paclitaxel and limus DCBs by Sedhom et al, including both de novo and ISR lesions, which associated the use of PCBs with better late angiographic outcomes, demonstrating lower rates of LLL along with higher rates of late lumen enlargement23. A significant limitation of that study is the combination of two distinct lesion types, de novo and ISR, within the same analysis. This approach obscures clear results, making it difficult to draw definitive conclusions, as these lesions have significantly different clinical and angiographic outcomes when treated with DCBs23. In our meta-analysis, in-segment LLL was slightly higher with PCBs compared with limus DCBs, despite similar in-lesion outcomes. This suggests that edge restenosis – potentially driven by paclitaxel’s short diffusion profile, which might cause less drug coverage at balloon edges and the drug “flake-off” phenomenon during catheter transit – may be more prominent with paclitaxel. In terms of ISR, on the other hand, among the studies included in our analysis, it was only the REFORM trial that failed to show non-inferiority for angiographic outcomes for biolimus DCBs compared with PCBs6. Possible factors explaining these discrepancies include the inclusion of both BMS and DES-ISR in the REFORM study, as well as variations in the quality and manufacturing process of the biolimus A9-coated balloon.
Aiming to provide more insight into this matter, the ongoing Sirolimus-Coated Balloon Versus Paclitaxel-Coated Balloon for the Treatment of Coronary In-Stent Restenosis trial (SIBLINT ISR; ClinicalTrials.gov: NCT04240444) will randomise 260 participants with ISR to receive either a sirolimus or a paclitaxel DCB. The primary endpoint of this study will be in-segment LLL at 9 months after the intervention.
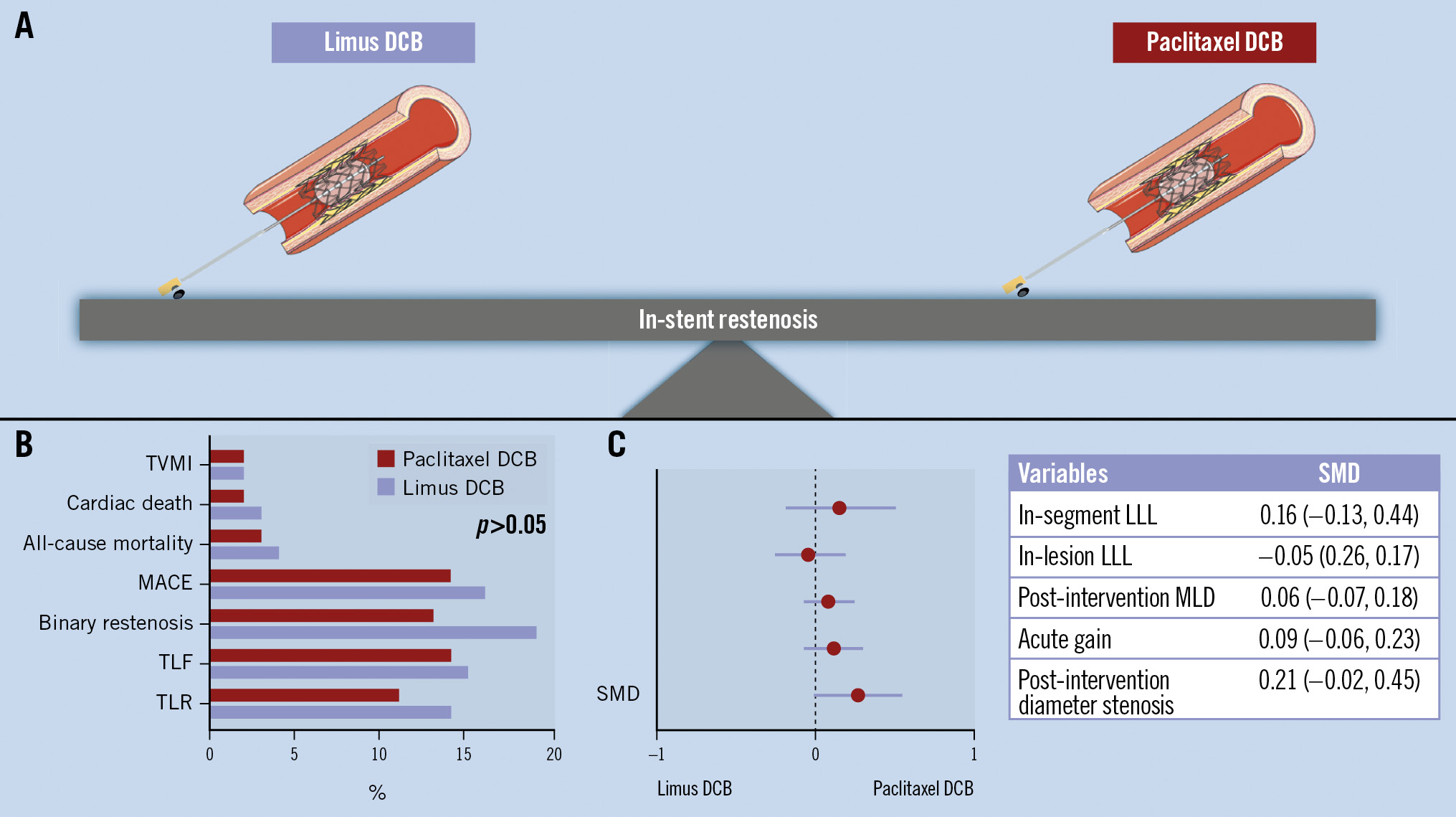
Central illustration. Comparison of limus- versus paclitaxel-coated balloons for treating ISR. A) Limus DCBs were equally effective as paclitaxel DCBs; (B) comparison of endpoints; (C) comparison of SMD between variables. Limus-coated balloons are as effective as paclitaxel-coated balloons for treating in-stent restenosis, demonstrating non-inferiority in both clinical and angiographic outcomes at 12 months following the intervention. DCB: drug-coated balloon; LLL: late lumen loss; MACE: major adverse cardiovascular events; MLD: minimal lumen diameter; SMD: standardised mean difference; TLF: target lesion failure; TLR: target lesion revascularisation; TVMI: target vessel myocardial infarction
Limitations
We acknowledge that our analysis is not free from certain limitations. First of all, a substantial number of high-risk patients with complex restenotic lesions were excluded from these trials. In addition to this, the methods utilised for lesion preparation are not thoroughly described in the included studies, though we assumed that all ISR lesions were optimally dilated before applying the DCBs. It was not possible to provide a robust comparative sensitivity analysis between sirolimus- and biolimus-coated balloons or drug-coating dosages due to the small sample size. Within the same context, the use of intravascular imaging modalities that enable detection of the mechanism of ISR and optimised lesion preparation before DCB application was inconsistent across the included studies. The total number of patients included in this meta-analysis was relatively low, a fact that, in combination with the low rate of clinical events, may also somewhat limit the statistical power of our results.
Conclusions
This meta-analysis of RCTs shows that limus-coated balloons are as effective as paclitaxel-coated balloons for treating ISR, demonstrating non-inferiority in both clinical and angiographic outcomes at 12 months following the intervention. We can observe that there is a trend for slightly better outcomes in the paclitaxel group in terms of luminology, however, without reaching statistical significance, considering the clinical outcomes at 12-month follow-up. These findings support the notion that both types of DCBs constitute an acceptable choice for the treatment of challenging ISR. There is still a need for future trials, though, to evaluate the effectiveness of new-generation DCBs and DES before establishing the best approach for the management of ISR.
Impact on daily practice
Drug-coated balloons (DCBs) have proven highly effective in treating in-stent restenosis (ISR). Our findings showed that there are no significant differences between paclitaxel-coated balloons (PCBs) and limus DCBs in the risk of major clinical outcomes, including all-cause mortality, cardiac death, and clinically driven target lesion revascularisation. Limus DCBs achieve at least equal angiographic outcomes compared with PCBs without significant differences in late lumen loss during 12-month follow-up. The study indicates that limus DCBs are a reliable alternative to PCBs in daily clinical practice for managing ISR. Future larger prospective studies might aim to validate these results for various indications.
Conflict of interest statement
The authors have no conflicts of interest to declare.
