Excimer laser coronary atherectomy (ELCA) to modify or debulk atherosclerotic plaque during percutaneous coronary intervention (PCI) has been in clinical use for the past four decades. Primarily, ELCA was introduced to mitigate the challenges posed by routine PCI for complex lesions and in crossing resistant lesions. It is also one of the preferred devices to debulk the soft neointimal hyperplastic tissue of in-stent restenosis (ISR) lesions12. However, ELCA use remains limited to a few operators; hence, its documented evidence is limited worldwide.
ELCA systems, such as the CVX-300 Excimer or Philips Laser System – Nexcimer (both Philips), generate laser energy through the interaction between chloride and xenon gases. This energy is then captured and conveyed via fibre optics to a catheter, which subsequently delivers it to the target lesion. ELCA works through three mechanisms: a light pulse, which modifies and breaks molecular bonds at a maximum depth of 50 micrometres; a sonic wave, which results from the release of energy, causing the microfracture of calcified and fibrotic material; and a cavitation bubble, which breaks soft tissue and enhances the lumen.
Since the introduction of ELCA into clinical use, PCI for complex lesions − such as uncrossable and undilatable lesions, calcified lesions, thrombotic lesions, and saphenous vein grafts − has demonstrated improved clinical outcomes345. Various operators have also demonstrated their utility for underexpanded stents and difficult-to-dilate calcified lesions67. Among the treatments for these complex lesions, ELCA has emerged and evolved significantly over the last few years8910. These evolutions include refinement of the console, improvement of the catheter, and technique modification, such as using saline as a medium for clearing the blood during lasing. These improvements have contributed to the efficacy and safety of this technology1112. Various clinical studies, such as CARMEL, the ELLEMENT registry, and the National Cardiovascular Data Registry (NCDR), have augmented the evidence of ELCA effectiveness for certain indications and established its safety1314151617. However, the overall data available are still limited in volume and diversity for application to real-world scenarios.
The reason for the low penetration of ELCA in aiding PCI is the apprehension regarding complications related to its use. Despite improvements in catheter design and procedural techniques, the adoption of ELCA has been limited. This is partly due to misunderstandings about how it works, the unsatisfactory initial experiences with the device, and a lack of awareness regarding the substantial safety and efficacy data that have been accumulated since those early trials. This study is the first to report the early experience of using ELCA in the Indian population. This study was designed to demonstrate the efficacy and safety of the ELCA device in the setting of complex PCI.
Methods
The present study represents the first single-centre, retrospective observational study of consecutive real-world patients undergoing PCI with ELCA. Consecutive patients requiring ELCA-assisted PCI who were treated between January 2021 and December 2023 at the Breach Candy Hospital, Mumbai, were included in the study. The primary objective of this study was to establish the safety and efficacy of ELCA in a real-world Indian population for various indications, including forms of coronary artery disease such as acute coronary syndrome (ACS) with a large thrombus burden, chronic total occlusions (CTOs), moderate to severe calcified lesions, and diffuse ISR. The indication for ELCA was at the physician’s discretion.
A complete list of study definitions is provided in Table 1.
Table 1. Definitions.
| Terms | Definitions |
|---|---|
| Major adverse cardiac events | Hierarchical composite of target lesion revascularisation, myocardial infarction, or cardiovascular death |
| Device success | For uncrossable lesions: |
| Successful crossing of the target lesion with the ELCA catheter | |
| Successful facilitation of the microcatheter by the ELCA | |
| For in-stent restenosis: | |
| Successful crossing of the index lesion with the catheter | |
| For thrombotic lesions: | |
| Establishment of TIMI 3 flow | |
| Technical success | Successful completion of the PCI procedure |
| Procedural complications | Procedure-related complications, major dissection, perforation requiring pericardial drainage, slow flow, no-reflow, haemodynamic instability requiring inotropic support, and chest pain with ECG changes persisting for over 20 mins |
| Escalating energy strategy | Gradually increasing energy from 45 J/mm2 at 40 Hz escalating to 80 J/mm2 at 80 Hz in a 0.9 mm catheter or 40 J/mm2 at 25 Hz escalating to 60 J/mm2 at 40 Hz in a 1.4 mm catheter |
| Direct maximum energy strategy | Maximum fluence and frequency: 80 J/mm2 at 80 Hz in a 0.9 mm catheter or 60 J/mm2 at 40 Hz in a 1.4 mm catheter |
| Uncrossable lesions | An uncrossable lesion was defined as a lesion that was successfully crossed with the wire, but where a microcatheter or a 1.0 mm balloon could not be advanced over it |
| ECG: electrocardiogram; ELCA: excimer laser coronary atherectomy; PCI: percutaneous coronary intervention; TIMI: Thrombolysis in Myocardial Infarction | |
Scientific and methodological assurance
As this was a retrospective observational study, to ensure that the study maintained scientific integrity and remained unbiased, a research protocol was prepared, reviewed, and approved by an ethics committee. The study data were recorded in a predesigned case report form, ensuring that all details were recorded accurately. This study complied with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines across four stages. First, all records were validated by comparing cath lab records with Philips billing records to verify data validity and accuracy. Second, the specified eligibility requirements were validated against hospital medical records. Third, all retrieved records were critically examined for the availability of follow-up data for predetermined timeframes of 30±10 days and 180±10 days post-procedure. Fourth, an independent reviewer assessed all the data and protocol compliance at each stage. The integrity and quality of the study findings depend heavily on this careful approach to data collection and reporting. The absence of dropouts and independent monitoring indicated the scientific integrity of the study. Among the 78 identified patients, none were excluded for eligibility, medical, or follow-up reasons (Figure 1).
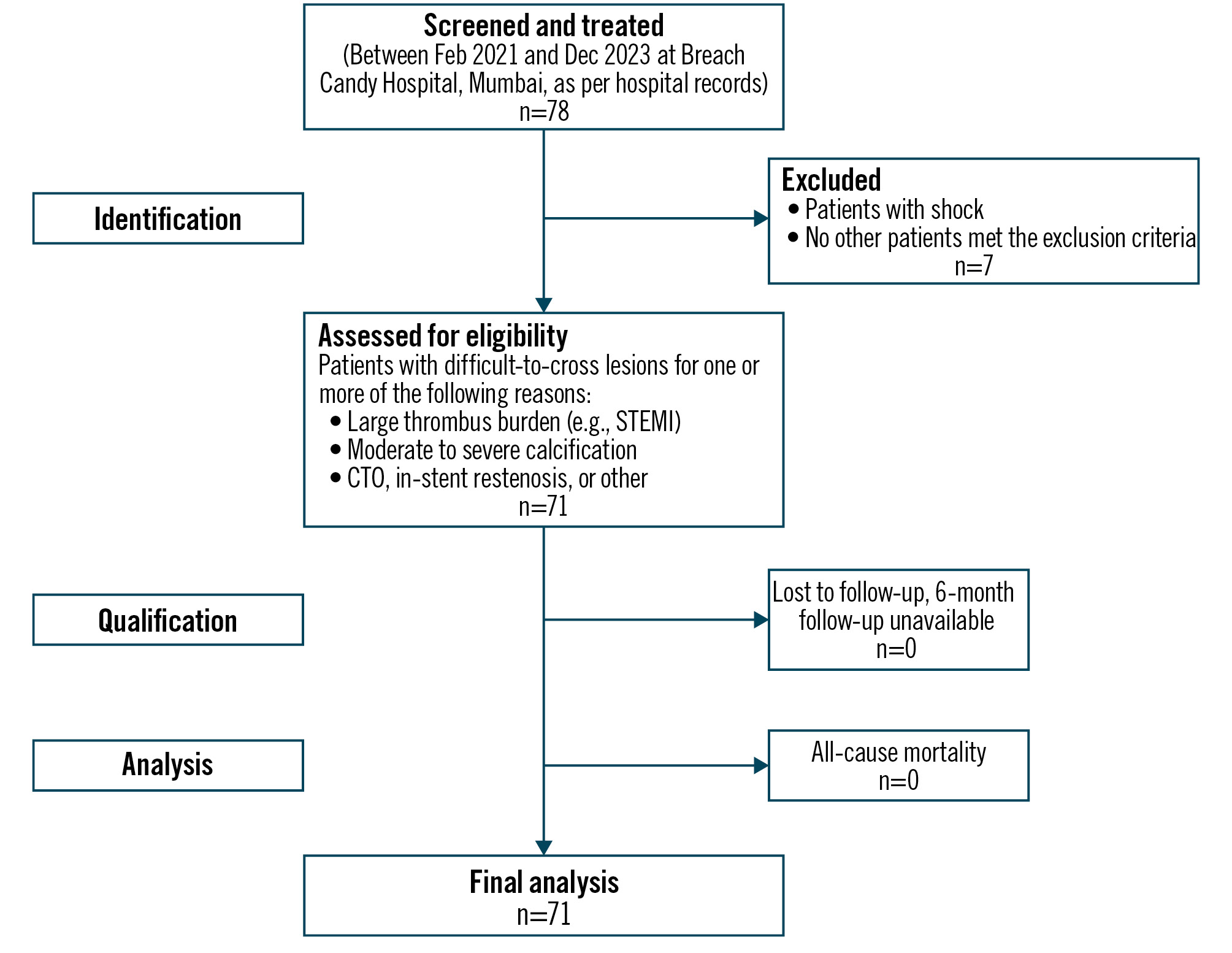
Figure 1. Flowchart for the subject disposition and analysis based on STROBE guidelines. CTO: chronic total occlusion; STEMI: ST-segment elevation myocardial infarction; STROBE: Strengthening the Reporting of Observational studies in Epidemiology
Statistical analysis and data collection
Data were collected using a predefined case report form and systematically prepared for evaluation. Statistical analysis was performed by an independent statistician at the Contract Research Organisation (Clicebo Solutions, New Delhi, India). All numerical data are reported as central tendency and spread, most commonly mean and standard deviation. All categorical data are reported as frequencies. For the analysis of statistical significance, p-values were calculated using McNemar’s test for data normalcy, the Student’s t-test for small datasets, and the z-test for large datasets. Considering the smaller spread of data distribution (single-centre study), the clinical effect size was calculated in terms of Cohen’s d. For applicability of the data in the wider population, in selected parameters, Cohen’s κ (kappa) was used for agreement analysis with the ULTRAMAN registry.
Results
Demographics and disease characteristics
This study included data from 71 consecutive patients at baseline, 30 days, and 6 months of follow-up. The data had a statistically significant normal distribution (McNemar p<0.001), indicating that the data were random and representative of real-world all-comers. The overall population included 62 (87.32%) males, with a mean age of 63.51±13.57 years. Hypertension was observed in 49 (69.01%) patients, diabetes in 36 (50.70%), dyslipidaemia in 39 (54.93%), while 5 patients (7.04%) were current smokers. Previous MI was noted in 7 patients (9.86%) and prior revascularisation in 32 patients (45.07%) (Table 2).
At baseline, stable coronary artery disease was present in 43 patients (60.56%), comprising 29 (51.95%) experiencing new angina and 14 (18.18%) with inducible ischaemia on a stress test. Acute coronary syndrome was present in 28 patients (39.44%), of whom 18 (25.35%) had ST-segment elevation myocardial infarction (STEMI) and 10 (14.08%) had non-STEMI (NSTEMI); the rest had chronic stable coronary artery disease and abnormal functional study. The mean left ventricular ejection fraction (LVEF) was 45.1±11.6%, with severe LV dysfunction (LVEF <30%) in 15 patients (21.13%). A total of 31 patients (43.66%) had a SYNTAX score >22, which is an indicator of high complexity of the coronary vasculature anatomy (Table 2).
Table 2. Demographics and medical history.
| Parameter | N=71 |
|---|---|
| Age | 63.51±13.57 |
| Sex | |
| Male | 62 (87.32) |
| Female | 9 (12.68) |
| Hypertension | 49 (69.01) |
| Diabetes | 36 (50.70) |
| Dyslipidaemia | 39 (54.93) |
| Current smoker | 5 (7.04) |
| Previous MI | 7 (9.86) |
| Previous CABG | 1 (1.41) |
| Previous PCI | 31 (43.66) |
| Chronic kidney disease | 7 (9.86) |
| Clinical presentation | |
| Acute coronary syndrome | 28 (39.44) |
| STEMI | 18 (25.35) |
| NSTEMI | 10 (14.08) |
| Stable coronary disease | 43 (60.56) |
| Stable angina | 29 (51.95) |
| Inducible ischaemia on stress test | 14 (18.18) |
| Low LVEF <30% | 15 (21.13) |
| Mean LVEF, % | 45.1±11.6 |
| SYNTAX score >22 | 31 (43.66) |
| Data are given as n (%) or mean±standard devation. p<0.001 for normal distribution (actual value 0.00000000106). The maximum effect size was Cohen’s d=2.38. CABG: coronary artery bypass grafting; LVEF: left ventricular ejection fraction; MI: myocardial infarction; NSTEMI: non-ST-segment elevation myocardial infarction; PCI: percutaneous coronary intervention; STEMI: ST-segment elevation myocardial infarction; SYNTAX: Synergy between percutaneous coronary intervention with Taxus and Cardiac Surgery | |
Angiographic characteristics
A total of 77 lesions were treated in 71 patients (mean 1.24 lesions/patient). The treated lesions involved the left main artery in 5 (6.49%), the left anterior descending artery in 36 (46.75%), the right coronary artery in 24 (31.68%), and the left circumflex artery in 12 (15.58%). The sample included complex lesions: multivessel PCI in 7 (9.09%), in-stent restenosis in 30 (38.96%), moderate to severely calcified lesions in 33 (42.86%), and 26 (33.77%) CTOs (Table 3).
Table 3. Angiographic characteristics.
| Angiographic characteristics | |
|---|---|
| Total number of lesions | 77 |
| Lesions per patient | 1.24 |
| ACS with thrombotic lesions | 27 (35.06) |
| Uncrossable CTO lesions | 26 (33.77) |
| Moderately to heavily calcified lesions | 33 (42.86) |
| Long lesions (>30 mm) | 50 (64.94) |
| Bifurcation lesions | 16 (20.78) |
| Long diffuse in-stent restenosis lesions | 30 (38.96) |
| Multivessel PCI | 7 (9.09) |
| Disease distribution by artery | |
| Left main | 5 (6.49) |
| Left anterior descending artery | 36 (46.75) |
| Left circumflex | 12 (15.58) |
| Right coronary artery | 24 (31.68) |
| Data are given as n or n (%). ACS: acute coronary syndrome; CTO: chronic total occlusion; PCI: percutaneous coronary intervention | |
Procedural details
Most PCIs are performed using radial or femoral access as the default. The decision to use ELCA was based on the operator’s assessment of the lesion: uncrossable, CTO/non-CTO, ISR, or STEMI/NSTEMI with a large thrombus burden. All procedures were performed using the CVX-300 Excimer laser system, with a 0.9 mm catheter in 68 patients (88.31%) and a 1.4 mm catheter in 8 patients (11.76%). Saline irrigation was used as the default medium for lasing in all patients, with escalation to contrast lasing medium in 7 patients (8.97%) according to procedural needs. Effective saline irrigation and blood clearance were achieved through the high concomitant use of guide extension catheters in proximity to the index lesion. The operators believe that this is an important step to reduce procedural complications.
The sample was observed to have a clear divergence over time between the escalating energy strategy in the early experience and the maximum energy setting as a default in the later period. An escalating energy strategy was used in the initial 23 lesions (32.36%) and a direct maximal energy strategy in 52 lesions (85.3%; see Table 1 for definitions). The pulses delivered were between 800 pulses and 8,576 pulses. The normality analysis of the pulse data confirms a systematic shift from the escalating energy strategy to the maximum energy setting as the default strategy (skewness: 1.58; kurtosis: 2.32) (Table 4).
ELCA was used to facilitate PCI as the first device by default for plaque modification or debulking. While ELCA enabled crossing of uncrossable lesions, lesion preparation strategies − such as cutting balloons in 41 lesions (53.25%), non-compliant balloons in 29 (37.66%), rotational atherectomy in 6 (7.79%), and intravascular lithotripsy (IVL) balloons in 9 (11.69%) − were used in a layered manner to facilitate final PCI. Intravascular imaging guidance was used for PCI in 49 patients (63.64%). PCI was completed using drug-eluting stents in 70 lesions (90.91%) and drug-eluting balloons in 8 lesions (10.39%). A guide extension catheter was used in 31 lesions (40.26%), only with the 0.9 mm catheters, to facilitate effective saline irrigation at the lesion site and better navigation through difficult proximal morphology (Table 5).
Table 4. ELCA details.
| Procedural characteristics | |
|---|---|
| Catheters used | 76 (100)# |
| Pulse count, NS | 2,563.5 [800, 8,576] |
| Catheter diameter | |
| 0.9 mm | 68 (88.31) |
| 1.4 mm | 8 (11.76) |
| Energy strategy | |
| Escalating energy strategy | 23 (32.36) |
| Maximum energy setting as a default | 52 (85.30) |
| Lasing medium | |
| Saline | 70 (89.74) |
| Contrast | 7 (8.97) |
| Data are given as n (%) or median [min, max]. #One patient required an escalation of catheter size from 0.9 mm to 1.4 mm. ELCA: excimer laser coronary atherectomy; NS: Newton seconds | |
Table 5. Additional devices used in the procedure.
| Variable | n=77 |
|---|---|
| Diagnostics | |
| IVUS | 49 (63.64) |
| OCT | 5 (6.49) |
| FFR | 4 (5.19) |
| Additional PCI procedures | |
| Stent | 70 (90.91) |
| Drug-eluting balloon | 8 (10.39) |
| Cutting balloon | 41 (53.25) |
| Non-compliant balloon | 29 (37.66) |
| Rotablator* | 6 (7.79) |
| IVL balloon | 9 (11.69) |
| Ancillary devices | |
| Guide extension catheter | 31 (40.26) |
| Data are given as n (%). *By Boston Scientific. FFR: fractional flow reserve; IVL: intravascular lithotripsy; IVUS: intravascular ultrasound; OCT: optical coherence tomography; PCI: percutaneous coronary intervention | |
Study outcomes
The study outcomes were assessed in two forms: ELCA procedure-oriented outcomes and patient-oriented outcomes. The procedure-oriented outcomes were assessed through the incidence of device success and procedural success, while patient-oriented outcomes were assessed based on the incidence of MACE at discharge, 1 month, and 6 months.
Device success and technical success
This study demonstrated a cumulative technical success rate of 100%. The overall ELCA device success rate was 96.10%. Despite ELCA failure in 3 (3.89%) previously uncrossable lesions, ELCA sufficiently modified the lesion and facilitated the use of the next layer of plaque modification techniques. PCI was performed for all lesions, achieving complete revascularisation.
The procedural success rate of ELCA was 85.71%, defined briefly as complication-free device success (p<0.005 [actual value 0.0037], effect size: Cohen’s d=–0.0778). A total of 12 procedural events (15.58%) were observed, attributed to main branch dissection in 5 (6.49%), chest pain with electrocardiogram changes lasting for 20 minutes or more in 1 (1.3%), side branch dissection or closure in 2 (2.6%), perforation in 1 (1.3%), and device failure in 3 (3.89%) (Table 6). The periprocedural events mentioned above were successfully managed using standard guidelines without any adverse events post-procedure.
Table 6. Procedural outcomes and success rates.
| Success | Percentage | |
|---|---|---|
| Cumulative technical success | 77 | 100 |
| Device success | 74 | 96.10 |
| Complete revascularisation (TIMI 3) | 68 | 95.77 |
| Procedural success | ||
| Complication-free device success | 66 | 85.71 |
| Procedural complications | ||
| Device failures | 3 | 3.89 |
| Main branch dissection | 5 | 6.49 |
| Chest pain ECG changes | 1 | 1.3 |
| S/B dissection/closure | 2 | 2.6 |
| Perforation | 1 | 1.3 |
| Data are given as n or %. N.B.: one event each was common among the following combinations: a. ECG changes, dissection, and side branch closure; b. dissection and side branch closure, dissection and technical failure. Three such overlapping incidents were counted only once at their latest hierarchical occurrence. ECG: electrocardiogram; S/B: side branch; TIMI: Thrombolysis in Myocardial Infarction | ||
Study outcomes
There were no intraprocedural deaths or major non-ischaemic complications. There were no unanticipated serious adverse events during any of the procedures.
Major adverse cardiac events
No MACE were reported during hospitalisation. At 1-month follow-up, the cumulative hierarchical MACE rate was 1 (1.41%), which was attributed to target lesion revascularisation. One (1.41%) additional target lesion revascularisation was reported between 1 and 6 months; thus, the cumulative hierarchical MACE rate at 6 months was 2 (2.82%; p<0.05) (Table 7, Central illustration).
Table 7. Outcomes – major adverse cardiac events.
| Parameter | Values | p-value | |
|---|---|---|---|
| n | % | ||
| In-hospital MACE | 0 | 0 | |
| MACE at 1 month | 1 | 1.41 | 0.13 |
| CV death | 0 | 0 | |
| MI | 0 | 0 | |
| TLR | 1 | 1.41 | 0.39 |
| MACE at 6 months (cumulative) | 2 | 2.82 | 0.04 |
| CV death | 0 | 0 | |
| MI | 0 | 0 | |
| TLR | 2 | 2.82 | 0.38 |
| CV: cardiovascular; MACE: major adverse cardiac events; MI: myocardial infarction; TLR; target lesion revascularisation | |||
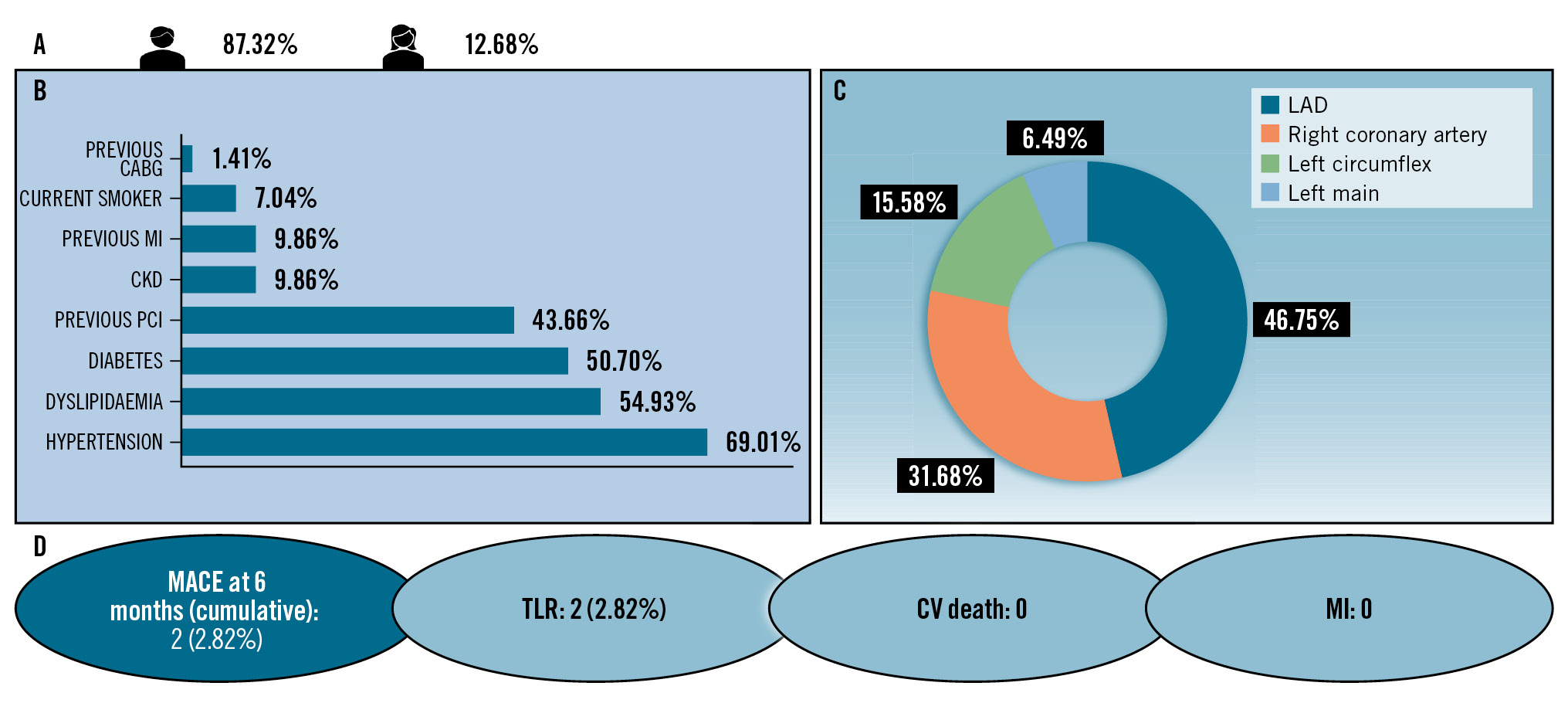
Central illustration. Real-world experience with ELCA: use of the CVX-300 Excimer laser system appeared to be a safe and effective strategy for PCI in complex lesions. A) Patient demographics; (B) comorbidities; (C) patterns of disease distribution; (D) cumulative MACE at 6 months. CABG: coronary artery bypass grafting; CKD: chronic kidney disease; CV: cardiovascular; ELCA: excimer laser coronary atherectomy; LAD: left anterior descending artery; MACE: major adverse cardiac events; MI: myocardial infarction; PCI: percutaneous coronary intervention; TLR: target lesion revascularisation
Discussion
Since its introduction in 2008, ELCA has registered success in difficult-to-treat lesions such as CTOs, ISR, severe calcification, and thrombotic lesions181920. In CARMEL, ULTRAMAN, and NDCR, ELCA-assisted PCI has shown greater success than the conventional plain old balloon angioplasty technique7202122. In their review article, Tsutsui et al explain the complexities and compiled common indications of ELCA2. Among these indications, balloon and microcatheter uncrossable lesions are particularly important. In these lesions, ELCA suitably modifies the plaque, rendering it crossable and thus suitable for PCI. In this study, ELCA was observed to have a positive effect on the outcome even when it appeared to have failed to cross the lesion, as it modified the plaque enough to allow the passage of a microcatheter across the lesions, which facilitated the advancement of the procedure. ELCA was established as an alternative effective technique for difficult-to-cross lesions, utilising a second layer of plaque modification techniques, including rotational atherectomy, high-pressure non-compliant balloons, and IVL balloons. Notably, the use of ELCA with additional devices produced high success rates, with no numerical differences despite thrombotic or calcified lesions, use of saline or contrast lasing medium, or use of the escalating energy approach or maximum energy as the default strategy. The ROLLERCOASTR-EPIC22 study evaluated the use of ELCA in calcified lesions and compared it with other devices such as Rotablator (Boston Scientific), IVL, and other laser technologies. The study showed a high crossover rate between arms, suggesting the synergistic use of these devices in calcified lesions23.
Evidence of the effectiveness of ELCA has been established across all specific subsets of difficult lesions. McQuillan et al registered 81.6% success in their study of 331 patients, with 473 of 637 lesions treated with ELCA. This study included 9.9% ISRs, 30.9% CTOs, and >49% uncrossable or undilatable lesions. Despite the complexity, there were only a few complications, including dissection (0.6%), no-reflow (0.6%), coronary perforation (2.7%), tamponade (0.2%), and perforation after laser catheter passage (0.6%)11. Li et al included 59 patients with difficult-to-cross lesions and compared ELCA and non-ELCA treatment. Lumen gain was higher in ELCA-assisted PCI (diameter stenosis 24.5±9.09% vs 35.1±18.6%; p=0.048 and minimal lumen diameter 2.36±0.29 mm vs 1.78±0.64 mm; p<0.001). The 9-month MACE rate was also lower in ELCA (9.5% vs 15.8%)19. The current study showed cumulative technical success in 100% of lesions and therefore appears to have equivalent outcomes.
The CARMEL multicentre study of 151 ACS patients reported 95% device success, 97% angiographic success, 91% overall procedural success, and 4% mortality. Complications included perforation (0.6%), dissection (5%), minor complications (3%), acute closure (0.6%), distal embolisation (2%), and bleeding (3%)16. The current study reported comparable outcomes with a low cumulative incidence of side branch closure or dissection (2.6%), main artery dissection (6.49%), perforation (1.3%), despite having high complexity lesions (SYNTAX score >22 in >40% of patients). Ambrosini et al published a study of 66 ACS patients with a 6-month event-free survival rate of 95%20. Farag et al published a study of 51 recurrent ISR patients with no in-hospital MACE, 11.8% at 12 months, and 23.5% in the long term (median 4 years). The technical and procedural success rates were reported to be 92% and 90%, respectively, with no ELCA-related coronary perforation13. Latib et al published the ELLEMENT registry of 28 undilatable lesions that were successfully dilated subsequent to ELCA; success was defined as a 1 mm gain in mean diameter or 20% improvement in residual diameter stenosis, with a 6.74% MACE rate14. Considering the complex population and difficult lesions, the complication and MACE rates in these studies are comparable with those in the current study, which had a 6-month MACE rate of 6.49%, mostly due to death within 1 month.
The 328 patients in ULTRAMAN included those with saphenous vein grafts (1.8%), ACS (53.4%), CTOs (5.5%), ISRs (32.4%), heavily calcified lesions (2.4%), and other difficult-to-treat lesions (4.6%). The ULTRAMAN study reported a >92.5% success rate for ELCA5. Despite the significant differences in baseline characteristics, the current study is at least equivalent to the ULTRAMAN study. There was similar technical success (ULTRAMAN >92.5% vs current study 100%) and MACE (ULTRAMAN 8.40% vs current study 2.82%). The overall outcomes of the current study were similar to those in the ULTRAMAN study.
Limitations
This study was based on an early, single-centre retrospective experience with ELCA in the Indian population. The data were retrospectively collected, and careful minimisation of bias was performed. However, despite these real-world findings, the widespread application of ELCA requires validation in a wider population.
Impact on daily practice
In complex lesions with difficult anatomy or lesion morphology, excimer laser coronary atherectomy (ELCA) can enable percutaneous coronary intervention (PCI) completion in rare cases of device failure. ELCA can be deployed with maximal energy as the default strategy, which is as safe as the escalating energy strategy and appears to be an effective approach for PCI of complex lesions. The outcomes of ELCA are favourable in the Indian population, with a low incidence of 6-month major adverse cardiac events.
Conclusions
The outcomes of this study clearly support the safety and efficacy of ELCA − even when using a maximum energy strategy as the default − as an alternative technique for PCI in complex, challenging lesions that are resistant to balloon angioplasty. The study also identified that ELCA, even in cases of device failure, ensures effective and complete PCI with additional plaque modification techniques that would otherwise not be usable.
Acknowledgements
The authors acknowledge Mr Varun Punamiya (a medical student) for his assistance in collecting and compiling the data. The authors also acknowledge Dr Ashish Indani for his analysis and authoring support. The authors further acknowledge Dr Priyadarshini Arambam from Clicebo Solutions, and Mr Mukul Singh and Mr Prateek Malviya from NIPER for their assistance with data analysis and publication writing support.
Funding
The study data were analysed and reported through an unconditional educational grant from Philips India.
Conflict of interest statement
The authors have no conflicts of interest to declare.
