The introduction of stents during percutaneous coronary intervention (PCI) aimed to address issues such as acute elastic recoil, the sealing of flow-limiting dissections, and preventing restenosis caused by the acute barotrauma associated with plain old balloon angioplasty1. New-generation drug-eluting stents (DES) have demonstrated safety and effectiveness in reducing device- and vessel-oriented adverse events and are endorsed by current guidelines. However, the use of permanent metallic implants comes with some potential drawbacks. Indeed, a constant risk of long-term stent-related complications (i.e., in-stent restenosis, stent thrombosis, and neoatherosclerosis) with an incidence rate of approximately 2-3% per stent per year has been described with current-era devices234.
The concept of an implant-free approach with drug-coated balloons (DCB) is particularly appealing in the context of small vessels, where PCI with DES is hampered by an inverse relationship between vessel diameter and future risk for restenosis356. In the setting of small vessels, DCB have been shown to be non-inferior compared with DES, both in terms of mechanistic and clinical outcomes in the short and long term78. Of note, the use of paclitaxel DCB (PCB) demonstrated signs of stabilisation in terms of the number of adverse events after 12 months, possibly related to the absence of permanent implants91011.
Clinical data from randomised controlled trials on DCB mainly focus on PCB. In contrast, modern DES release sirolimus or its analogues, as these have shown superior outcomes compared to paclitaxel-eluting stents12. While sirolimus provides enhanced safety, efficacy, and greater antirestenotic and anti-inflammatory effects than paclitaxel when used in coronary stents, its application in DCB is hampered by its low lipophilicity and limited penetration and retention in the target vessel wall. To date, sirolimus-coated balloon (SCB) technologies have provided conflicting results1314. Overall, as with PCB, a class effect is not expected, and whether some platforms outperform others is still under investigation.
The PICCOLETO VI study sought to compare the angiographical and physiological outcomes after treatment of de novo coronary artery disease (CAD) with different PCB and SCB technologies.
Methods
Study population
This is an international, multicentre, investigator-driven, retrospective study that included 293 unselected patients from 17 high-volume centres that were expert in DCB angioplasty for de novo lesions. Patients were enrolled between May 2014 and July 2023 and had follow-up angiography available. Each investigator involved in the study had to certify adequate experience in DCB PCI, namely the use of at least 30 DCB for de novo lesions per year in the previous 5 years. Eligible patients presented with a clinical indication to PCI (stable CAD or acute coronary syndrome) and underwent angioplasty with an SCB or a PCB for de novo lesions. Exclusion criteria were creatinine clearance <30 mL/min, left ventricular ejection fraction <30%, life expectancy <12 months, as well as culprit lesion stenosis >99% and/or Thrombolysis in Myocardial Infarction (TIMI) flow <2, visible thrombus at the lesion site, concomitant or previous PCI of the same vessel with a stent, bifurcation lesions where side branch treatment was anticipated, left main stem disease, and ST-segment elevation myocardial infarction. Patients were scheduled to undergo an elective coronary angiography at 5-9 months after the index procedure.
The study was conducted in accordance with the ethical standards set by the Declaration of Helsinki and was approved by the ethics committees of each participating hospital. Written informed consent was obtained from all patients for anonymous data collection.
Coronary intervention
Invasive coronary angiography and PCI were performed in accordance with the local standard of care, via radial or femoral access, and following consensus documents. The strategy of lesion preparation with non-compliant, semicompliant, or cutting balloons was left to the operator’s discretion, as well as the decision to perform intravascular imaging pre- and/or post-PCI. Bailout stenting was discouraged unless DCB PCI evolved into a flow-limiting dissection (TIMI flow grade <3) or provoked acute vessel recoil after a minimum of 5 minutes from drug application. The antithrombotic regimen was left to the operator’s choice, but a minimum of 30 days of dual antiplatelet therapy was suggested in case of stable CAD. In the case of acute coronary syndrome or bailout stenting, a regimen of 6 to 12 months was indicated.
Study devices
A total of nine distinct DCB technologies were included, in accordance with the standard practices of the respective centre. PCB technologies were SeQuent Please NEO (B. Braun), ELUTAX 3 (Aachen Resonance), Prevail (Medtronic), Pantera Lux (Biotronik), RESTORE (Cardionovum), and AGENT (Boston Scientific). SCB technologies were MagicTouch (Concept Medical), SELUTION (MedAlliance), and SeQuent SCB (B. Braun). The pressure and inflation times of the balloon were applied as recommended by the manufacturers. The balloon length was recommended to exceed the lesion at least 2 mm proximally and distally. If needed, multiple DCB were allowed.
Coronary angiogram assessment
Angiography was performed before and after PCI and at the elective angiographic follow-up using identical projections. Coronary angiograms were anonymised, then sent to be centrally analysed in an independent core laboratory (Ferrara University Hospital, Ferrara, Italy). Quantitative coronary angiography (QCA) analysis and computation of the single-view Murray law-based quantitative flow ratio (μFR) were performed using quantitative flow ratio software (AngioPlus Core, version V3 [Pulse Medical]) by an experienced analyst, who was blinded to the antiproliferative drug and device used.
QCA analysis included the evaluation of reference vessel diameter (RVD), minimal lumen diameter (MLD), percentage diameter stenosis (%DS), and lesion length (LL). Acute gain (MLD post-DCB treatment–MLD at baseline), late lumen loss (LLL; MLD at 6-month follow-up–MLD post-DCB treatment), and net lumen gain (MLD at 6-month follow-up–MLD at baseline) were also computed. Late lumen enlargement (LLE) was considered a negative LLL. Binary restenosis was defined as detecting a 50% degree of stenosis in the target segment at follow-up.
μFR analysis was performed at baseline (pre-PCI), at the end of the index procedure, and at the elective angiographic follow-up. Fiducial colocalisation was applied, using side branches as markers. Exclusion criteria were ostial disease in the left main or in the right coronary artery and poor angiographic image quality. The detailed methodology for single-view μFR computation has been described previously and is reported in Supplementary Appendix 1151617. The standard cutoff value of μFR (≤0.80) was used to classify the haemodynamic significance18.
Late functional loss was defined as the paired difference of μFR values between post-procedure and follow-up, net functional gain as the paired difference of μFR values between pre-procedure and follow-up, and acute functional gain as the paired difference of μFR values between pre- and post-procedure.
ENDPOINT DEFINITIONS
The primary physiological endpoint was to compare the late functional loss among PCB and SCB platforms, defined according to the DCB Academic Research Consortium (ARC) criteria19. The coprimary endpoint of the study was to compare the late lumen loss in the two types of platforms. Secondarily, angiographic (net lumen gain and rate of binary restenosis) and physiological (net functional gain, μFR, and rate of ischaemia-inducing vessels) variables were compared between devices. Target lesion failure (TLF) was defined as the composite occurrence of cardiovascular death, target vessel myocardial infarction, and clinically driven target lesion revascularisation.
Statistical analysis
The sample size of this retrospective cohort study was calculated to be powered for non-inferiority of the primary endpoint at the angiographic follow-up, derived from the PICCOLETO II μFR study20. A mean±standard deviation late functional loss of –0.02±0.06 was expected with PCB. Using a non-inferiority margin of 0.06 and assuming an attrition rate of 10%, with a ratio of 2:1 between PCB and SCB, an overall cohort of 230 patients was required to achieve 90% power to demonstrate non-inferiority with a 1-sided type 1 error of 0.05. Categorical variables are reported as a percentage and were compared with the χ2 test or Fisher’s exact test, as appropriate. A 2-sided p-value<0.05 was considered statistically significant. Continuous variables are presented as mean and standard deviation if normally distributed and were compared with an unpaired t-test. Hazard ratios with 95% confidence intervals (CI) are provided. Receiver operating characteristic (ROC) curve analysis was performed and the area under the curve (AUC) was calculated to assess and compare the ability of the postprocedural μFR to predict ischaemic μFR values (≤0.80) at follow-up. Sensitivity, specificity, diagnostic accuracy, and the optimal cutoff value were defined from the calculated ROC curve.
All analyses were performed with SPSS Statistics, version 26 (IBM). Graphics were generated with GraphPad Prism, version 7.0 (Dotmatics).
Results
Baseline characteristics
After applying exclusion criteria and having discarded incomplete or unanalysable angiographic data among 293 patients (293 vessels), a total of 227 patients (for a total of 227 coronary lesions) underwent QCA and assessment of μFR at baseline and follow-up. These were treated either with a PCB (n=148: ELUTAX 3, n=34; Prevail, n=28; SeQuent Please NEO, n=68; Pantera Lux, n=9; RESTORE, n=5; Agent, n=4) or with an SCB (n=79: MagicTouch, n=51; SELUTION, n=25; SeQuent SCB, n=3).
Baseline clinical characteristics were comparable between the PCB and SCB groups: patients were primarily males (77.5%; 72.9% vs 86.6%; p=0.09) with a mean age of 66±9 years (66±9 years vs 68±9 years; p=0.06). Clinical characteristics are reported in Table 1.
Table 1. Baseline clinical and procedural characteristics.
| Overall n=227 | PCB n=148 (65.2) | SCB n=79 (34.8) | p-value | |
|---|---|---|---|---|
| Baseline characteristics | ||||
| Male | 176 (77.5) | 108 (72.9) | 68 (86.6) | 0.09 |
| Age, years | 66±9 | 66±9 | 68±9 | 0.06 |
| Hypertension | 180 (79.4) | 118 (79.7) | 62 (78.5) | 0.84 |
| Diabetes | 84 (37.2) | 51 (34.7) | 33 (41.7) | 0.34 |
| Smoker | 49 (21.6) | 37 (25.0) | 12 (15.2) | 0.07 |
| Dyslipidaemia | 165 (72.8) | 111 (75.0) | 54 (68.4) | 0.38 |
| PAD | 21 (9.0) | 11 (7.4) | 10 (12.7) | 0.26 |
| Previous MI | 70 (30.7) | 54 (36.3) | 16 (20.3) | 0.03 |
| Previous CABG | 7 (3.4) | 6 (4.7) | 1 (1.3) | 0.23 |
| Previous PCI | 92 (40.7) | 63 (42.7) | 29 (36.7) | 0.45 |
| LVEF, % | 53.1±10.4 | 53.4±9.8 | 52.8±11.3 | 0.80 |
| Stable CAD | 142 (62.4) | 81 (54.4) | 61 (77.2) | 0.02 |
| Procedural characteristics | ||||
| Radial access | 213 (93.9) | 137 (92.6) | 76 (96.1) | 0.18 |
| Bifurcations | 76 (33.3) | 46 (31.5) | 30 (37.9) | 0.52 |
| Ostial lesion | 34 (15.4) | 16 (10.8) | 18 (22.8) | 0.02 |
| Severe calcification | 54 (23.8) | 41 (27.8) | 13 (17.1) | 0.08 |
| Mdc, mL | 189±85 | 194±81 | 182±89 | 0.41 |
| Predilatation | 218 (96.1) | 145 (98.1) | 73 (93.1) | 0.08 |
| Max size of predilatation balloon, mm | 2.5±0.6 | 2.4±0.6 | 2.5±0.5 | 0.44 |
| DCB size, mm | 2.6±0.5 | 2.6±0.6 | 2.6±0.5 | 0.84 |
| DCB inflation, atm | 9.5±3.3 | 8.9±3.3 | 10.4±3.2 | 0.01 |
| DCB length sum, mm | 32.6±18.8 | 33.3±18.6 | 31.6±19.1 | 0.55 |
| IVUS/OCT | 58 (25.6) | 46 (31.1) | 12 (15.2) | 0.03 |
| Non-flow-limiting dissections | 27 (11.8) | 25 (16.9) | 2 (2.6) | 0.15 |
| Bailout stenting | 4 (1.8) | 3 (2.0) | 1 (1.3) | 0.54 |
| Device success | 223 (98.3) | 144 (97.3) | 79 (100) | 0.15 |
| Values are n (%) or mean±standard deviation. CABG: coronary artery bypass graft; CAD: coronary artery disease; DCB: drug-coated balloon; IVUS: intravascular ultrasound; LVEF: left ventricular ejection fraction; Mdc: contrast media; MI: myocardial infarction; OCT: optical coherence tomography; PAD: peripheral artery disease; PCB: paclitaxel-coated balloon; PCI: percutaneous coronary intervention; SCB: sirolimus-coated balloon | ||||
Procedural characteristics
Intracoronary imaging was used more frequently in PCB-based PCI (31.1% vs 15.2%; p=0.03). No further procedural differences were found between the PCB and SCB groups. The radial approach was predominant and was used in 93.9% of cases (n=213). It was observed that 15.4% (n=34) involved ostial lesions and that one-third of the cases were bifurcation lesions (n=76). In most cases, the lesions had been predilated (96.1%), and the incidence of non-flow-limiting dissections at the final result was evidenced in 11.8% (n=27). Device success was achieved in 98.3% of the procedures and bailout stenting was needed in only 4 cases. Procedural characteristics are reported in Table 1.
Pre-PCI QCA revealed no statistically significant differences between the groups (Table 2). The RVD was 2.2±0.6 mm (2.3±0.6 mm vs 2.2±0.6 mm; p=0.56), MLD 0.80±0.48 mm (0.83±0.53 mm vs 0.75±0.36 mm; p=0.26) and %DS 65.4±17.2% (64.7±18.5% vs 66.9±14.6%; p=0.36). Moreover, the mean LL was 23.5±14.8 mm for the overall sample (22.6±11.6 mm vs 25.2±19.4 mm; p=0.22).
Baseline μFR values were comparable between PCB and SCB (0.52±0.29 vs 0.47±0.28; p=0.18). Most lesions were classified as flow-limiting (μFR ≤0.80; n=186, 81.9%), with no difference between PCB and SCB (80.4% vs 84.8%; p=0.41) (Table 3).
Table 2. Quantitative coronary analysis results.
| Overall n=227 | PCB n=148 (65.2) | SCB n=79 (34.8) | p-value | |
|---|---|---|---|---|
| Pre-PCI | ||||
| RVD, mm | 2.2±0.6 | 2.3±0.6 | 2.2±0.6 | 0.56 |
| MLD, mm | 0.80±0.48 | 0.83±0.53 | 0.75±0.36 | 0.26 |
| DS, % | 65.4±17.2 | 64.7±18.5 | 66.9±14.6 | 0.36 |
| Lesion length, mm | 23.5±14.8 | 22.6±11.6 | 25.2±19.4 | 0.22 |
| Post-PCI | ||||
| MLD, mm | 1.55±0.54 | 1.55±0.58 | 1.56±0.46 | 0.84 |
| DS, % | 35.1±11.9 | 34.2±12.4 | 36.8±10.8 | 0.11 |
| Acute gain, mm | 0.76±0.56 | 0.72±0.62 | 0.82±0.45 | 0.19 |
| Follow-up (5 months [IQR 3-7]) | ||||
| MLD, mm | 1.56±0.58 | 1.61±0.58 | 1.46±0.56 | 0.07 |
| DS, % | 36.0±14.3 | 33.5±13.9 | 40.8±13.9 | 0.01 |
| Net gain, % | 0.76±0.66 | 0.77±0.67 | 0.72±0.63 | 0.51 |
| Late lumen loss, mm | –0.01±0.57 | –0.05±0.56 | 0.10±0.59 | 0.05 |
| Positive remodelling | 118 (52.0) | 86 (58.1) | 32 (40.5) | 0.01 |
| In-segment restenosis | 27 (11.9) | 13 (8.8) | 14 (17.7) | 0.05 |
| Values are mean±standard deviation or n (%). DS: diameter stenosis; IQR: interquartile range; MLD: minimal lumen diameter; PCB: paclitaxel-coated balloon; PCI: percutaneous coronary intervention; RVD: reference vessel diameter; SCB: sirolimus-coated balloon | ||||
Table 3. Murray law-based quantitative flow ratio.
| Overall n=227 | PCB n=148 (65.2) | SCB n=79 (34.8) | p-value | |
|---|---|---|---|---|
| Pre-PCI | ||||
| Vessel μFR | 0.50±0.29 | 0.52±0.29 | 0.47±0.28 | 0.18 |
| Vessel μFR ≤0.80 | 186 (81.9) | 119 (80.4) | 67 (84.8) | 0.41 |
| Post-PCI | ||||
| Vessel μFR | 0.87±0.11 | 0.86±0.13 | 0.87±0.08 | 0.41 |
| Vessel μFR ≤0.80 | 36 (15.8) | 26 (17.6) | 10 (12.7) | 0.34 |
| Acute functional gain | 0.36±0.28 | 0.33±0.28 | 0.40±0.27 | 0.08 |
| Follow-up (5 months [IQR 3-7]) | ||||
| Vessel μFR | 0.86±0.13 | 0.87±0.13 | 0.85±0.14 | 0.27 |
| Net functional gain | 0.35±0.28 | 0.34±0.28 | 0.37±0.30 | 0.39 |
| Late functional loss | 0.01±0.14 | –0.01±0.15 | 0.03±0.13 | 0.09 |
| Negative loss | 118 (51.9) | 85 (57.4) | 33 (41.8) | 0.02 |
| Vessel μFR ≤0.80 | 43 (18.9) | 22 (14.9) | 21 (26.6) | 0.03 |
| Values are n (%) or mean±standard deviation. IQR: interquartile range; PCB: paclitaxel-coated balloon; PCI: percutaneous coronary intervention; SCB: sirolimus-coated balloon; μFR: Murray law-based quantitative flow ratio | ||||
Immediate post-PCI outcomes
Immediately following PCI, no significant differences between PCB and SCB were observed in terms of acute angiographic (0.72±0.62 mm vs 0.82±0.45 mm; p=0.19) or functional (+0.33±0.28 vs +0.40±0.27; p=0.08) gain. Post-PCI μFR values were comparable between the two groups (0.86±0.13 vs 0.87±0.08; p=0.41), as were the proportions of ischaemia-inducing lesions (17.6% vs 12.7%; p=0.34).
Angiographic follow-up
Elective angiographic follow-up was performed at a median time of 5 months (interquartile range: 3-7). LLL was lower in the PCB group than in the SCB group (–0.05±0.56 mm vs +0.10±0.59 mm; p=0.05). Additionally, both groups showed LLE, albeit with a significantly higher proportion in the PCB group (58.1% vs 40.5%; p=0.01). The incidence of binary restenosis was lower in the PCB cohort compared with the SCB cohort (8.8% vs 17.7%; p=0.01). Cumulative frequency distributions of MLD, %DS, angiographic LLL, and net gain are reported in Figure 1 and Supplementary Figure 1.
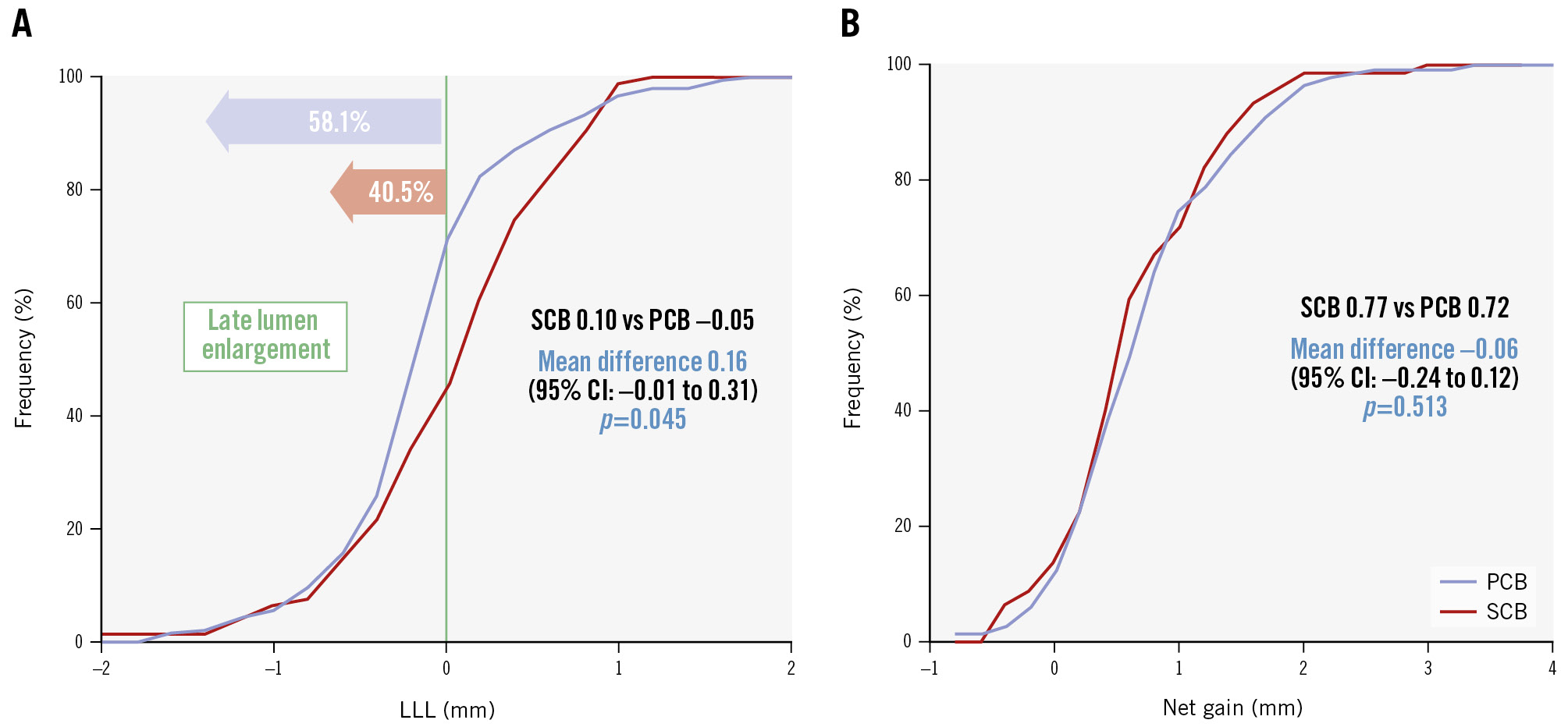
Figure 1. Cumulative frequency distribution of late lumen loss and net gain. A) The cumulative frequency distribution curve of late lumen loss; (B) the cumulative frequency distribution curve of net gain. CI: confidence interval; LLL: late lumen loss; PCB: paclitaxel-coated balloon; SCB: sirolimus-coated balloon
Physiological follow-up
Late functional loss − the study’s primary endpoint − did not differ between PCB and SCB (−0.01±0.15 vs +0.03±0.13; p=0.09), nor did net functional gain (+0.34±0.28 vs +0.37±0.30; p=0.39). Crude μFR values were also comparable (0.87±0.13 vs 0.85±0.14; p=0.27), while a higher rate of ischaemia-inducing vessels at follow-up was detected in the SCB group (14.9% vs 26.6%; p=0.03).
Cumulative frequency distributions of μFR, late functional loss, and net functional gain are reported in Figure 2. Late functional loss did not differ significantly among different PCB and SCB technologies, as shown in Figure 3. The detailed subgroup interaction analysis for late functional loss is presented in Supplementary Figure 2.
At the median clinical follow-up time of 9 months, TLF occurred in 4.7% of the vessels, with no significant difference between PCB and SCB (3.4% vs 6.8%; p=0.279). Kaplan-Meier survival curves for target vessel failure and/or ischaemia at follow-up according to DCB type are reported in Supplementary Figure 3.
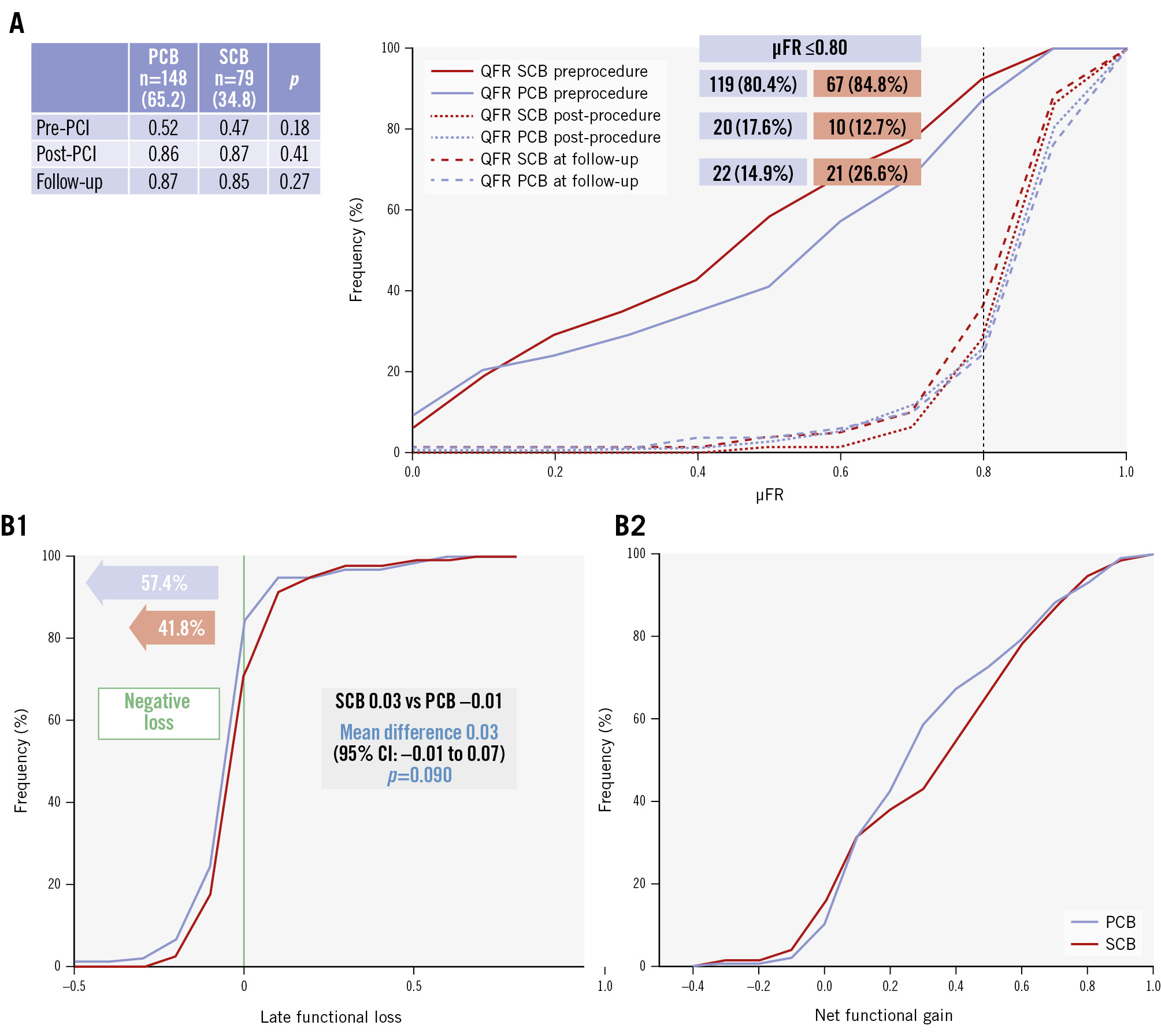
Figure 2. Murray law-based QFR at 5-month follow-up and cumulative frequency of late functional loss and net gain. A) The cumulative frequency distribution of μFR pre-PCI, post-PCI, and at follow-up. The table shows mean values and t-test comparison. The rate of ischaemic vessels (μFR ≤0.80) in the two groups is reported in the boxes. B1) The cumulative frequency distribution curve of late functional loss; (B2) the cumulative frequency distribution curve of net functional gain. CI: confidence interval; PCB: paclitaxel-coated balloon; PCI: percutaneous coronary intervention; QFR: quantitative flow ratio; SCB: sirolimus-coated balloon; μFR: Murray law-based QFR
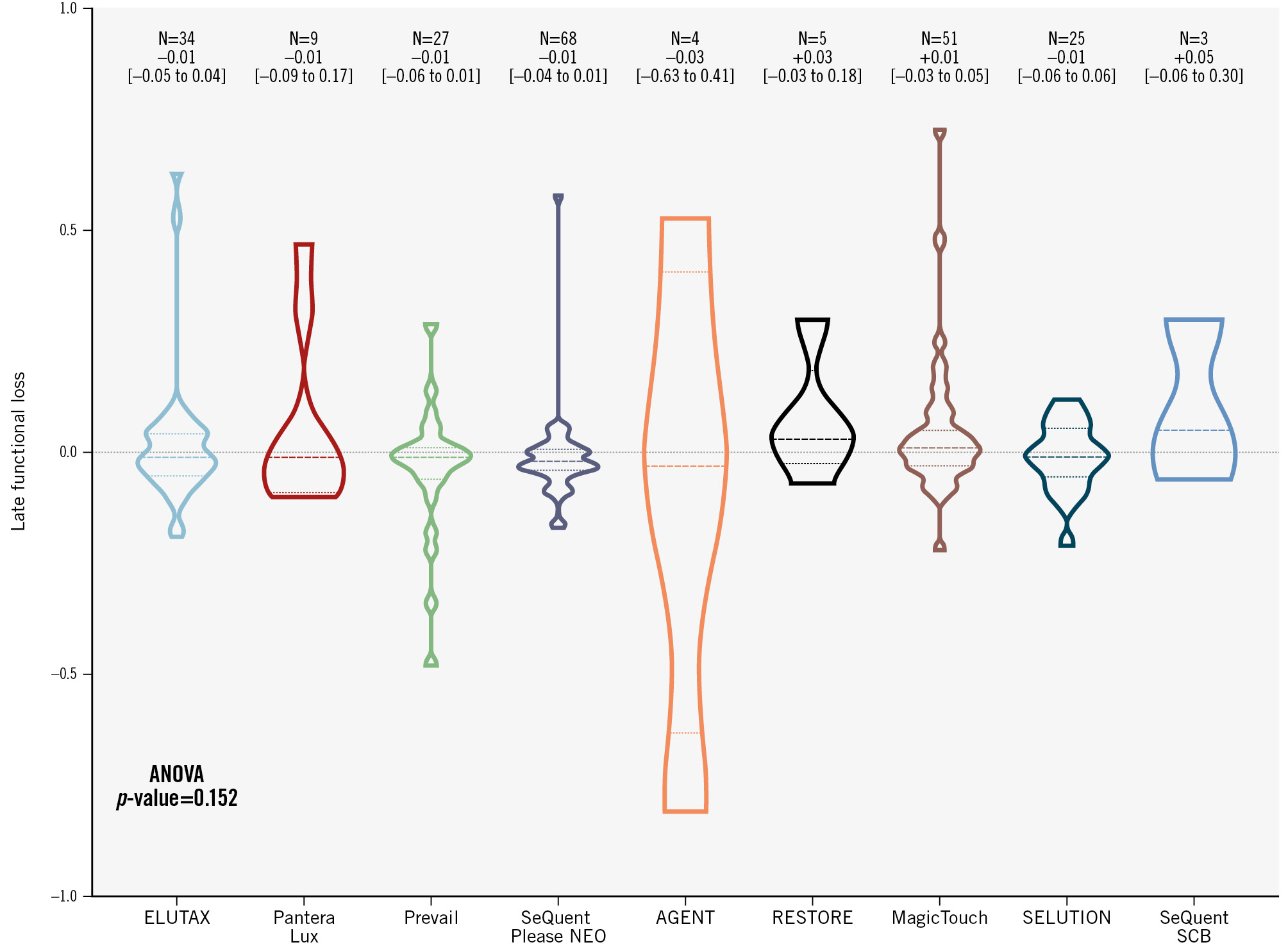
Figure 3. Comparison of late functional loss among different PCB and SCB technologies. ANOVA: analysis of variance; PCB: paclitaxel-coated balloon; SCB: sirolimus-coated balloon
Post-PCI physiology and ischaemia at follow-up
Post-PCI μFR was seen to significantly interact with the detection of ischaemic μFR values (≤0.80) at follow-up. In ROC curve analysis (Figure 4A), the AUC of μFR in predicting a follow-up μFR ≤0.80 was 0.758 (95% CI: 0.697-0.812; p<0.001), with no difference between PCB and SCB (p=0.712).
According to the AUC, the optimal μFR cutoff value after DCB angioplasty to predict follow-up ischaemia was a μFR ≤0.86, with an accuracy of 81.1%, a sensitivity of 74.4%, a specificity of 69.0%, a negative predictive value (NPV) of 92.0%, and a positive predictive value (PPV) of 36.0%. Vessels with a post-DCB μFR ≤0.86 presented a higher rate of ischaemic μFR values at follow-up, as compared to vessels with a post-DCB μFR>0.86 (38.2% vs 9.2%; p<0.001), as displayed in Figure 4B.
The QCA-defined recommended post-PCI cutoff value (%DS ≥30%) did not interact with the detection of ischaemic μFR values (≤0.80) at follow-up. In ROC curve analysis (Figure 4A), the AUC of %DS in predicting a follow-up μFR ≤0.80 was 0.525 (95% CI: 0.443-0.605; p=0.677), with a significant difference compared to post-PCI μFR (p=0.001).
Post-PCI %DS ≥30% provided a sensitivity of 71.1%, a specificity of 22.8%, an NPV of 71.1%, and a PPV of 22.9%. There was no significant difference in terms of ischaemic μFR values at follow-up in vessels with a post-DCB %DS ≥30% or %DS <30% (20.6% vs 15.2%; p=0.337), as displayed in Figure 4B.
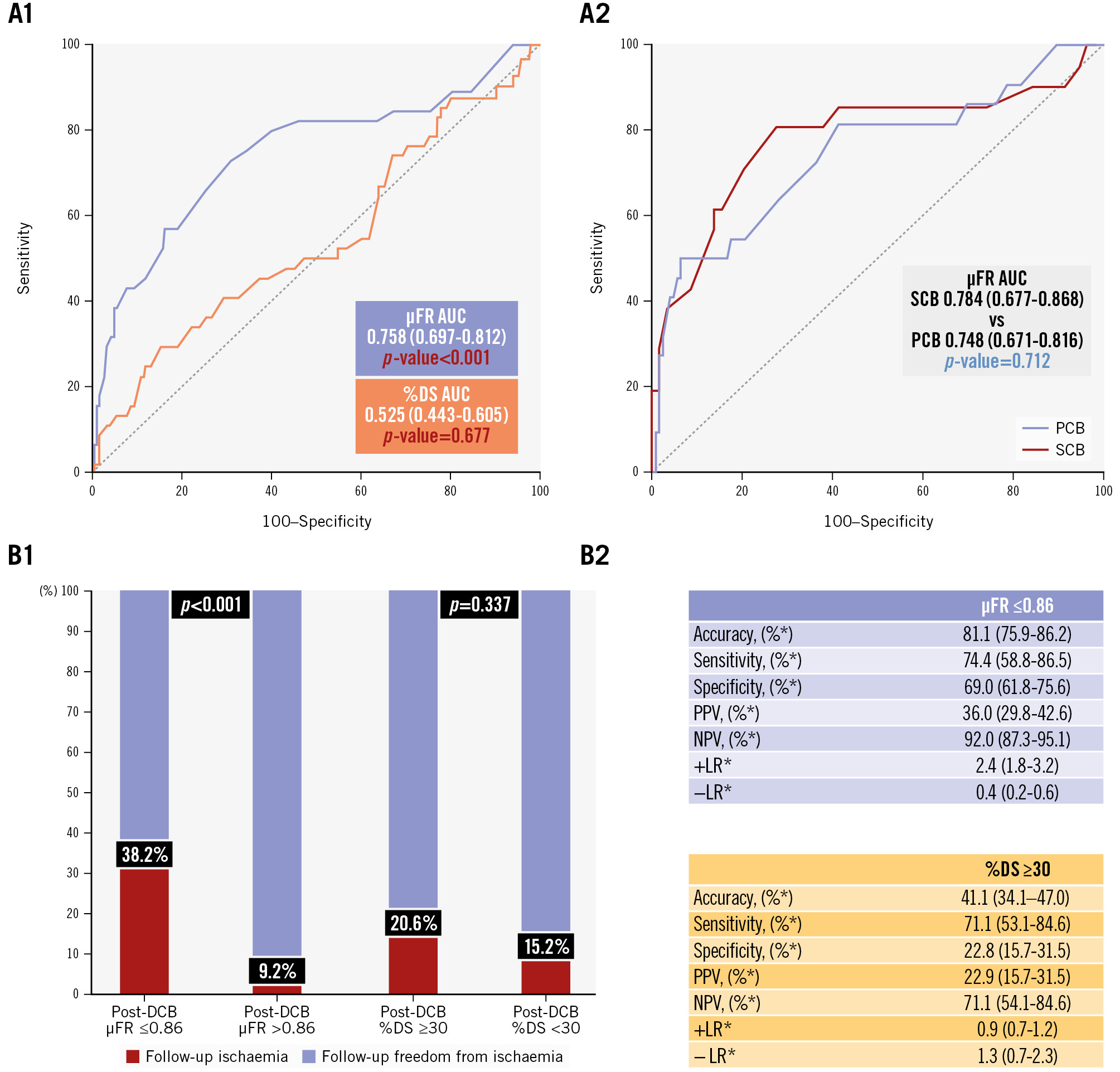
Figure 4. μFR and QCA-%DS value interaction with follow-up ischaemia (μFR ≤0.80) after DCB treatment. A) Receiver operating characteristic curves for predicting follow-up ischaemia: (A1) diagnostic performance of post-DCB μFR (purple) and quantitative coronary angiography-%DS (orange) in predicting follow-up ischaemia; (A2) diagnostic performance of post-DCB μFR in predicting follow-up ischaemia, stratified by paclitaxel-coated balloons (purple) and sirolimus-coated balloons (red). AUC and 95% CI are given. Diagnostic performance of post-DCB μFR ≤0.86 (B1) and quantitative coronary angiography %DS ≥30% (B2) in predicting follow-up ischaemia (red). *Percentages are given with 95% CI. AUC: area under the curve; CI: confidence interval; DCB: drug-coated balloon; %DS: percentage diameter stenosis; LR: likelihood ratio; NPV: negative predictive value; PCB: paclitaxel-coated balloon; PPV: positive predictive value; SCB: sirolimus-coated balloon; μFR: Murray law-based quantitative flow ratio
Discussion
The PICCOLETO VI study provides an in-depth comparative analysis of various PCB and SCB for the treatment of de novo CAD at expert centres, highlighting mechanistic insights regarding both angiographic and physiological outcomes. The study showed comparable immediate and late functional performance between PCB and SCB, with PCB demonstrating superior angiographic remodelling parameters (Central illustration).
Paclitaxel is currently the most commonly used antiproliferative drug in DCB therapy. However, sirolimus presents potential advantages in terms of safety and efficacy due to its broader therapeutic range and strong antirestenotic and anti-inflammatory effects, which partially contributed to the discontinuation of paclitaxel-eluting stent use in coronary arteries. Additionally, recent debatable safety concerns surrounding PCB, such as risks of cytotoxicity and distal embolisation, have raised the possibility of using an alternative cytostatic drug for DCB therapy21. Of note, PCB have been shown to be safe both in coronary and peripheral arteries in large patient-level meta-analyses1022.
Our study found that late functional loss was not significantly different between the two therapies, whereas, as in previous studies, the PCB group showed more favourable angiographic outcomes, with lower late lumen loss (–0.05 vs +0.10; p=0.05) and a higher prevalence of LLE (57.4% vs 41.8%; p=0.01). Moreover, PCB-treated vessels were less likely to be associated with physiological ischaemia at follow-up, as compared to those treated with SCB (14.9% vs 26.6%; p=0.03).
This finding aligns with previous studies that have demonstrated the effectiveness of paclitaxel in reducing restenosis rates, particularly in small vessel disease, where the risk of restenosis is typically higher because of the established inverse relationship between target vessel segment diameter and restenosis at follow-up3523. Moreover, our findings regarding the comparison of PCB and SCB are in line with previous experiences. In the TRANSFORM I randomised clinical trial including small vessel de novo CAD, this difference was primarily attributed to the significantly lower LLL (0.00 vs 0.32; p<0.001) observed with the PCB (SeQuent Please NEO) compared with the SCB (MagicTouch), with LLE at follow-up occurring in 53.7% and 30.0% of the cases, respectively13. Similarly, according to Ahmad et al in a study of 70 patients with de novo CAD in non-small vessels randomised to treatment with either an SCB (SeQuent SCB) or a PCB (SeQuent Please), LLE at the 6-month angiographic follow-up was observed in nearly two-thirds of lesions treated with a PCB and about one-third of those treated with an SCB14. Such findings were further confirmed with the same technologies in the European population, with comparable LLL (0.04 vs 0.11; p=0.440) and LLE (56.3% vs 44.1%; p=0.544) rates24. Historically, the rate of positive remodelling reported with plain balloon angioplasty ranged from 10% to 20%25. The favourable LLL with PCB can be attributed to its unique pharmacokinetic properties; paclitaxel, being lipophilic, quickly crosses cell membranes, binds to microtubules, and inhibits cell division and migration, thus preventing cell proliferation. On the other hand, distal embolisation has been reported in preclinical data after PCB treatment, while it has never emerged with any of the available SCB technologies26. Of note, such a phenomenon has not shown any clinical impact in clinical trials so far. The recent ANDROMEDA patient-level meta-analysis revealed that for de novo small vessel disease, treatment with PCB PCI is associated with a lower rate of major adverse cardiac events and a comparable rate of TLF at 3 years when compared to DES implantation11. Moreover, while it failed to show non-inferiority in the overall population, PCB were non-inferior to modern DES in the small-vessel subgroup in the recent REC-CAGEFREE I trial27.
Our cohort included predominantly small vessels (average RVD 2.2 mm), slightly larger than in TRANSFORM I (RVD 2.05 mm), while Ahmad et al and Scheller et al included non-small vessels (RVD 2.8 mm and 2.9 mm, respectively). Notably, our real-world study included diffusely diseased vessels with an average lesion length at QCA analysis of 24 mm, and nearly half of the vessels had a lesion length greater than 20 mm. In randomised controlled trials (RCT), the median lesion length ranges from 10 mm in TRANSFORM I to 14 mm in PICCOLETO II and BELLO, with consequently longer DCB used during PCI (33 mm on average in our sample vs 20-24 mm in the other trials), also reflecting the expertise of the centres involved in this study781328. This different lesion setting potentially led to lower physiological values at the elective angiographic follow-up (overall μFR 0.86; PCB 0.87 vs SCB 0.85) compared with TRANSFORM I (overall μFR 0.89; PCB 0.91 vs SCB 0.86) or the RESTORE SVD China subanalysis (PCB μFR 0.89)29.
Despite the angiographic advantages observed with PCB, the overall clinical outcomes, as measured by TLF at 9 months, did not significantly differ between the two groups. Although the study was not powered for event detection, this observation suggests that angiographic improvements may not necessarily translate into better physiological or clinical outcomes. The broader context of these results aligns with findings from previous large-scale studies. A recent meta-analysis including 1,861 patients showed no significant difference in TLF between PCB and SCB (odds ratio 1.01, 95% CI: 0.75-1.35), despite the angiographic advantage of PCB (LLL −0.11, 95% CI: –0.23 to +0.02)30. Similar findings were highlighted in the 2,123 patients enrolled in the EASTBOURNE study, the largest prospective study on SCB, where the rates of TLR were 2% and 2.6% at 1 and 2 years in de novo lesions, respectively3132. Several mechanisms may underlie this observation. Differences in drug kinetics between paclitaxel and sirolimus may partly explain this finding: paclitaxel’s rapid tissue uptake and prolonged retention favour early antiproliferative effects and positive remodelling, whereas sirolimus acts more slowly, promoting uniform healing but less immediate lumen gain. In addition, PCB-induced remodelling may improve vessel geometry without fully restoring microvascular or diffuse flow, and μFR, though validated, may underestimate subtle haemodynamic improvements in such settings, contributing to the observed dissociation between angiographic and physiological results.
This study is also the first to highlight the predictive value of post-DCB physiology for detecting ischaemia at follow-up, defining μFR ≤0.86 as the optimal threshold. The international DCB and the DCB ARC consensus documents recommend a residual stenosis post-DCB treatment of %DS ≤30%, estimated visually, as the optimal threshold for successful balloon angioplasty193334. However, our analysis found that this cutoff did not correlate with long-term ischaemia and demonstrated a significantly lower predictive performance compared to μFR (AUC 0.758 vs 0.525; p=0.001). This suggests that μFR is a valuable tool in assessing the functional success of DCB interventions and could potentially guide postprocedural management to improve long-term outcomes. While the goal of PCI is to restore normal coronary haemodynamics, suboptimal physiology is common even after angiographically “successful” PCI, with post-PCI fractional flow reserve (FFR) ≤0.90 in nearly half of cases and ≤0.80 in about 10%2. Multiple studies and meta-analyses have demonstrated that suboptimal post-PCI physiology is associated with worse outcomes3536. Despite these data, routine physiological guidance remains limited because of procedural and cost barriers; consequently, most interventions still rely on angiography alone1837. Relying solely on a single angiographic view, μFR is an improved version of angiography-derived FFR that is conceived to overcome the impact of linear vessel tapering that might otherwise hamper the reliability of QFR in assessing bifurcations stenoses; μFR provides an artificial intelligence-empowered delineation of vessel contours, frame counting, and more accurate reconstruction of reference vessel size, with a fast and reproducible computation, all of which reduce operator dependence on the measurement1517. Nonetheless, given the retrospective design of the present study, the proposed μFR ≤0.86 threshold should be considered hypothesis-generating and requires validation in larger, prospective, randomised studies to confirm its predictive value and clinical applicability within a physiology-guided DCB PCI strategy.
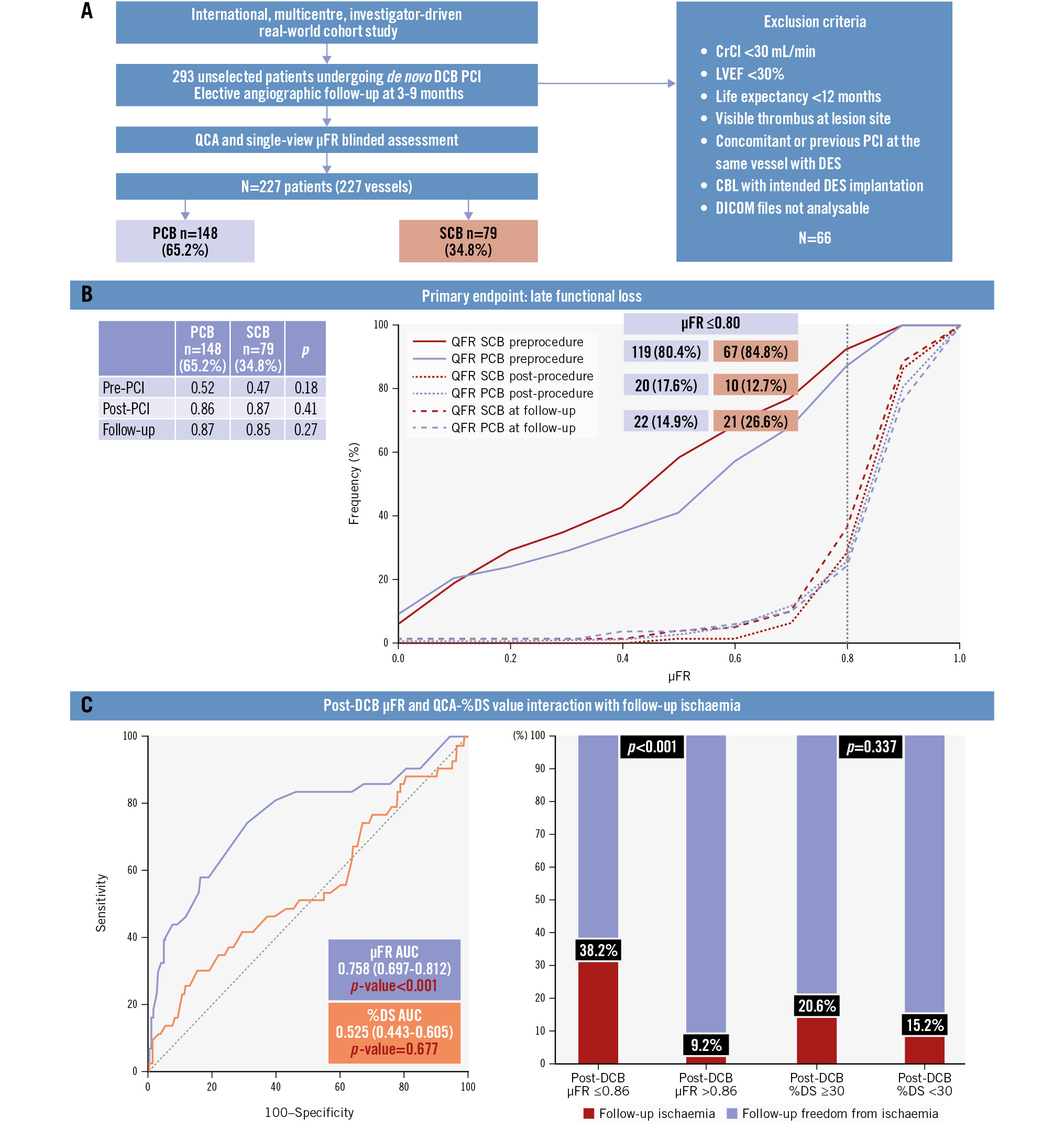
Central illustration. Angiographic and functional assessment after paclitaxel or sirolimus DCB for de novo lesions of small coronary vessels: PICCOLETO VI study. A) Selection flowchart; (B) primary endpoint of late functional loss; (C) post-DCB treatment μFR and QCA-%DS value interaction with follow-up ischaemia. %DS: percentage diameter stenosis; AUC: area under the curve; CBL: coronary bifurcation lesion; CrCl: creatinine clearance; DCB: drug-coated balloon; DES: drug-eluting stent; DICOM: Digital Imaging and Communications in Medicine; LVEF: left ventricular ejection fraction; PCB: paclitaxel-coated balloon; PCI: percutaneous coronary intervention; QCA: quantitative coronary angiography; QFR: quantitative flow ratio; SCB: sirolimus-coated balloon; μFR: Murray law-based QFR
Limitations
While the PICCOLETO VI study provides valuable insights, several limitations should be acknowledged. First, the retrospective, non-randomised design introduces inherent selection bias and potential confounding factors, particularly given the inclusion of multiple DCB platforms with different drug formulations and delivery technologies. Although independent and blinded analyses were performed, such heterogeneity may partly account for the observed differences between devices. Second, although adequately powered for physiological and angiographic analyses, the study was not designed to detect small differences in clinical outcomes, especially for relatively infrequent events such as TLF. The relatively short follow-up period also limits the assessment of long-term durability and the clinical relevance of the observed physiological and angiographic benefits. Third, differences in operators’ experience, lesion preparation techniques, and local device availability across centres may have influenced procedural results, despite standardised inclusion criteria and independent quantitative analysis. Future studies should therefore aim for prospective, randomised, head-to-head comparisons of different DCB technologies with standardised procedural protocols to isolate the mechanical and clinical effects of each platform.
Conclusions
In conclusion, the PICCOLETO VI study provides new mechanistic and physiological insights into DCB angioplasty, showing comparable immediate and late functional performance between PCB and SCB, with PCB demonstrating superior angiographic remodelling parameters. Given the retrospective and heterogeneous nature of the study, these findings should be interpreted as hypothesis-generating. Larger, prospective, randomised investigations are warranted to confirm these observations and to define more precise, physiology-based criteria for optimal DCB selection and use in clinical practice.
Impact on daily practice
Paclitaxel-coated balloons (PCB) have strong supporting evidence for use in small coronary vessels, while sirolimus-coated balloons have shown variable angiographic results, raising questions about their overall effectiveness. PICCOLETO VI confirms the angiographic superiority of PCB but also shows that late functional loss is comparable between platforms. The identification of a Murray law-based quantitative flow ratio of ≤0.86 after drug-coated balloon (DCB) treatment as a reliable predictor of ischaemia at follow-up suggests a practical, operator-independent marker to guide optimisation and early risk stratification. These findings support integrating physiology into DCB selection and evaluation, while underscoring the need for larger randomised studies to clarify long-term outcomes and the role of physiology-guided DCB strategies.
Funding
Core laboratory activities were supported by a donation from the Fondazione Ricerca e Innovazione Cardiovascolare.
Conflict of interest statement
S. Fezzi reports a grant from Science Foundation Ireland (15/RP/2765); and reports speaker honoraria from and consultancy for Boston Scientific, Shockwave Medical, and Teleflex. T.T. Rissanen reports consultancy for Boston Scientific, Abbott, Cordis, Biotronik, and B. Braun. F. Ribichini reports research grants from Philips and Abbott. The other authors have no conflicts of interest to declare relevant to the contents of this paper.
